PharmaCare Newsletter
April 2026 PharmaCare Newsletter
Print off the PDF of this PharmaCare Newsletter, and post it in your pharmacy for staff to read!
April 2026 PharmaCare Newsletter (PDF)
Find past issues on the newsletter search page.

The current edition of PAD Refills is an ADHD drug table update. Make sure to subscribe so you don’t miss out on news and updates!
PharmaCare expands coverage to diabetes devices as part of national pharmacare agreement
PharmaCare has expanded coverage to two hybrid closed-loop (HCL) systems with support from national pharmacare funding. The Omnipod 5 was listed on April 1, 2026, and Ypsomed AG’s mylife YpsoPump was listed on December 17, 2025. Both are limited coverage benefits, requiring Special Authority (SA) approval.
An HCL is a type of automated insulin delivery (AID) system that links or “loops” an insulin pump and a glucose monitor, using algorithmic software to automatically adjust insulin delivery according to glucose data. HCL pumps do not automatically give people access to looping functions. A separate glucose monitor that pairs with the pump, to enable looping functions, is required. For coverage, separate SA approval of the glucose monitor is required.
| HCL-capable pump Limited coverage, SA required for all pumps |
Glucose monitor Limited coverage, SA required for all glucose monitors |
Compatible insulins | |||
| 100% coverage, Plan NP | Non-benefits | ||||
| mylife Ypsopump | Dexcom G6 and FreeStyle Libre 3 Plus | Admelog®, Apidra® | NovoRapid®, Humalog® | ||
| Omnipod 5 | Dexcom G6 and G7 | Admelog®, Trurapi®, Kirsty® | NovoRapid®, Humalog® | ||
Omnipod 5 and mylife Ypsopump have 100% coverage under Plan B, Plan C, Plan F and Plan W, and income-based coverage under Fair PharmaCare, with coverage depending on deductible and family maximum requirements. They are not covered under Plan NP.
Other technologies in this treatment space are under review. PharmaCare will consider additional evidence-informed changes to increase access to diabetes devices over the term of the national pharmacare agreement with Health Canada.
Resources
- Diabetes devices and supplies PINs
- SA for Insulin Pumps
- Limited coverage criteria – Dexcom Continuous Glucose Monitors (CGMs)
- Limited coverage criteria – FreeStyle Libre Flash Glucose Monitors (FGMs)
- BC PharmaCare expands coverage of diabetes-related supplies
Changes to CGM and FGM SA adjudication, initial approval periods
Effective April 1, 2026, Special Authority (SA) coverage requests for continuous glucose monitors (CGMs) and flash glucose monitors (FGM) will be adjudicated through the normal review process, and no longer automatically. At the same time, initial SA approval periods have been extended from 1 year to 3 years. Limited coverage criteria are unchanged.
The change ensures CGM and FGM coverage aligns with the limited coverage criteria. A recent PharmaCare review found that a high proportion of CGM and FGM SA requests were approved even though the patient did not meet eligibility requirements. In some cases, PharmaCare revoked SA coverage while processing a renewal application. Loss of coverage can be distressful for patients and add to prescribers’ administrative burden. As well, providing SA coverage for ineligible patients impacts the sustainability of the PharmaCare program.
Reviewing coverage requests through the usual review process will reduce disruptive coverage reversals. The extended initial approval period will mean fewer renewal applications, lessening administrative burden, and support continuity of care for patients.
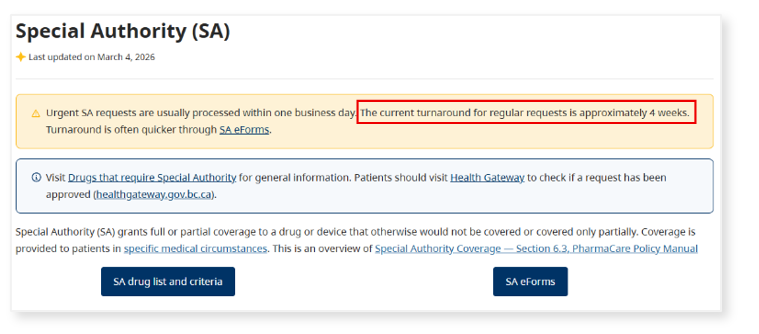
While CGM and FGM coverage decisions will take longer – reviews of non-urgent SA requests currently take 4 weeks on average – the approach supports long-term sustainability of PharmaCare while maintaining patient access to medically necessary therapies. The Ministry of Health is exploring digital solutions to make adjudication processes more efficient.
The Special Authority (SA) webpage is regularly updated with approximate turnaround times for SA requests. Response times are often quicker through SA eForms.
Clients can track the status of their SA requests through Health Gateway.
Resources
PharmaCare covers orbital prostheses as of April 1
As of April 1, 2026, PharmaCare covers custom-manufactured orbital prostheses for people who have lost eyelids and/or surrounding anatomy due to disease or injury. B.C. residents are eligible for coverage of one right-side and/or one left-side orbital prosthesis every three years. PharmaCare will cover up to a maximum of $8,500 per side.
PharmaCare now also covers facial retouching services for ear, nose, and orbital prostheses. Coverage for retouching is limited to once every 365 days, up to a maximum of twice per lifetime of each prosthesis. Additional retouch services may be considered on an exceptional basis as needed.

Pre-approval through an anaplastologist registered as a PharmaCare provider is required. Before the prostheses or retouching services are received, clients must register with Fair PharmaCare, and/or have coverage under Plan B (Long-term Care), Plan C (Income Assistance and Family Services), or Plan F (Children in the At-Home Program). Coverage under Fair PharmaCare is subject to deductible or family maximum requirements.
For more details, visit the PharmaCare Prosthetic and Orthotic Policy Manual, Section 5.4: Custom ear and nose prostheses and services. To apply for coverage, submit HLTH 5404 – PharmaCare Prosthetic Benefits (Non-Limb) Application for Financial Assistance. Review PharmaCare’s Facial prostheses PINs.
Resources
- Prosthetic and Orthotic Policy Manual, Section 5.4: Custom ear and nose prostheses and services
- HLTH 5404 – PharmaCare Prosthetic Benefits (Non-Limb) Application for Financial Assistance
- Facial prostheses PINs
Medical Laboratory Week – April 12-18, 2026
Medical Laboratory Week will be celebrated in B.C. and throughout Canada from April 12 to April 18, 2026, to recognize laboratory professionals and their critical role in the health care system. This year’s theme is “Every patient, every test, every stage of life.”
Medical laboratory professionals work to support a wide range of medical professionals and practices, working in sample collection, analysis, and results delivery. Laboratory professionals play a critical role in supporting accurate diagnoses – laboratory results inform over 70% of clinical decisions.
Common laboratory terms and definitions
Phlebotomist: Trained healthcare professional who specializes in collecting blood specimens safely and accurately, ensuring patient comfort, proper labeling, and adherence to infection‑control standards.
Specimen collection: Process of obtaining biological samples—such as blood, urine, or saliva—for laboratory analysis. Proper specimen collection is essential to ensure accurate test results.
Venipuncture: Medical procedure of puncturing a vein, typically in the arm, using a sterile needle to collect blood. Venipuncture is the most common method used in phlebotomy.
Centrifugation: Laboratory process that uses rapid spinning to separate blood components (such as plasma, serum, and cells) based on density. Centrifugation is a key preparatory step before many diagnostic tests.
Pre‑analytical phase: All processes that occur before laboratory testing, including patient preparation, specimen collection, labeling, handling, and transport.
Post‑analytical phase: All processes that occur after testing is completed, including result validation, reporting, interpretation, communication of critical values, and record retention.
Visit Canadian Society for Medical Laboratory Science to participate in the lab decoration contest and for a list of landmarks across Canada that will be lit up in indigo colours to celebrate Medical Laboratory Week.
Take a moment during Medical Laboratory Week to thank medical laboratory professionals for their commitment and dedication!
Resources
- National Medical Laboratory Week – Canadian Society for Medical Laboratory Science
- Medical Laboratory Professionals Week – British Columbia Society of Laboratory Science
Policy Spotlight: Special Authority

Resources
- PharmaCare Policy Manual, Section 6.3: Special Authority Coverage
- gov.bc.ca/pharmacaresadruglist – list of drugs requiring SA approval
- gov.bc.ca/specialauthorityeforms – SA eForms
- healthgateway.gov.bc.ca – Health Gateway
Formulary and listing updates
Limited Coverage benefits: sotatercept (Winrevair®), denosumab biosimilars (Stoboclo™ and Osenvelt™), zilucoplan (Zilbrysq™), rosanolixizumab (Rystiggo®), tocilizumab (Avtozma™), and Omnipod® 5 automated insulin delivery (AID) system
PharmaCare has added the following limited coverage items to the PharmaCare drug list. Special Authority approval is required for coverage.
| Drug name | sotatercept (Winrevair®) | ||
|---|---|---|---|
| Date | March 11, 2026 | ||
| Indication | For the treatment of adults with pulmonary arterial hypertension (PAH), World Health Organization (WHO) Group 1 and Functional Class (FC) II or III | ||
| DINs | 02551306 02551314 |
Strength & form | 45 mg/vial in 1-vial or 2-vial kit 60 mg/vial in a 1-vial or 2-vial kit |
| Notes | When entering claims for sotatercept, the unit of measurement is vials, as per Correct quantities for PharmaCare claims. | ||
| Drug name | denosumab biosimilars (Stoboclo™ and Osenvelt™) | ||
|---|---|---|---|
| Date | March 25, 2026 | ||
| Indication | Stoboclo: For the treatment of osteoporosis (limited coverage benefit) Osenvelt: Hypercalcemia of malignancy (Plan P) |
||
| DINs | Stoboclo: 02560917 Osenvelt: 02560895 |
Strength & form | Stoboclo: 60 mg/mL pre-filled syringe Osenvelt: 120 mg/1.7 mL single-use vial |
| Notes | Osenvelt is a PharmaCare benefit under Plan P (Palliative Care) only. When entering claims for denosumab, the unit of measurement is volume in millimetres, as per Correct quantities for PharmaCare claims. |
||
| Drug name | zilucoplan (Zilbrysq™) | ||
|---|---|---|---|
| Date | March 25, 2026 | ||
| Indication | For the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-acetylcholine receptor (AChR) antibody positive | ||
| DINs | 02549220 02549239 02549247 |
Strength & form | 16.6 mg/0.416 mL single-dose pre-filled syringe 23 mg/0.574 mL single-dose pre-filled syringe 32.4 mg/0.81 mL single-dose pre-filled syringe |
| Notes | When entering claims for zilucoplan, the unit of measurement is the number of syringes, as per Correct quantities for PharmaCare claims. | ||
| Drug name | rozanolixizumab (Rystiggo®) | ||
|---|---|---|---|
| Date | March 25, 2026 | ||
| Indication | For treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive | ||
| DIN | 02556081 | Strength & form | 280 mg/2 mL single-dose vial |
| Notes | When entering claims for rozanolixizumab, the unit of measurement is the number of vials, as per Correct quantities for PharmaCare claims. | ||
| Drug name | tocilizumab (Avtozma™) | ||
|---|---|---|---|
| Date | April 1, 2026 | ||
| Indication | For the treatment of: | ||
| DINs | 02562022 02562030 02562049 02562057 02562065 |
Strength & form | 80 mg/4 mL single-use vial for intravenous infusion 200 mg/10 mL single-use vial for intravenous infusion 400 mg/20 mL single-use vial for intravenous infusion 162 mg/0.9 mL pre-filled syringe for subcutaneous injection 162 mg/0.9 mL autoinjector for subcutaneous injection |
| Drug name | Omnipod® 5 automated insulin delivery (AID) system (or hybrid closed-loop system) | ||
|---|---|---|---|
| Date | April 1, 2026 | ||
| Indication | For the management of diabetes mellitus | ||
| PINs | Omnipod 5 automated insulin delivery (AID) system starter kit: 45230022 | ||
| Omnipod 5 pods: 46340041 (insulin pump pod) | |||
| Compatible glucose monitor(s) | Dexcom G6 and G7 (PharmaCare limited coverage benefits) |
||
| Compatible insulins | Admelog®, Trurapi®, Kirsty® (Plan NP benefits with 100% coverage) |
||
| NovoRapid®, Humalog® (PharmaCare non-benefits) | |||
Effective April 1, 2026, Insulet’s Omnipod 5 automated insulin delivery (AID) system has been added as a limited coverage benefit. The Omnipod EROS and DASH remain PharmaCare benefits.
The Omnipod 5 AID system can be paired with either the Dexcom G6 or G7 continuous glucose monitor to create a hybrid-closed loop system that offers automated insulin delivery. The starter kit will be available to PharmaCare-covered patients at a cost of $6,300 and the pods are priced at $360 per box of 10 pods. Fair PharmaCare coverage is subject to annual deductible and family maximum amounts.
Non-benefits: mepolizumab (Nucala®), ravulizumab (Ultomiris®)
PharmaCare has decided not to cover the following drugs for the noted indication.
| Drug name | mepolizumab (Nucala®) | ||
|---|---|---|---|
| Date | March 11, 2026 | ||
| Indication | Severe chronic rhinosinusitis with nasal polyps | ||
| DINs | 02492989 02492997 |
Strength & form | 100 mg/mL solution in a pre-filled autoinjector for subcutaneous injection 100 mg/mL solution in a pre-filled syringe for subcutaneous injection |
| Drug name | ravulizumab (Ultomiris®) | ||
|---|---|---|---|
| Date | March 25, 2026 | ||
| Indication | For the treatment of adult patients with anti-acetylcholine receptor (AChR) antibody-positive generalized myasthenia gravis (gMG) | ||
| DINs | 02491559 02533448 02533456 |
Strength & form | 300 mg/30 mL (10 mg/mL) single-dose vial 300 mg/3 mL (100 mg/mL) single-dose vial 1100 mg/11 mL (100 mg/mL) single-dose vial |
EDRD benefit: etranacogene dezaparvovec (Hemgenix®)
PharmaCare has initiated funding of the following drug through the Expensive Drugs for Rare Diseases (EDRD) process. Clinicians may apply for funding for eligible patients as described below.
| Drug name | etranacogene dezaparvovec (Hemgenix®) | ||
|---|---|---|---|
| Date | March 16, 2026 | ||
| Indication | For treatment of adults (aged 18 years of age or older) with hemophilia B (congenital Factor IX deficiency) who require routine prophylaxis to prevent or reduce the frequency of bleeding episodes | ||
| DIN | 02542560 | Strength & form | 1 × 1013 genome copies/mL, suspension for intravenous infusion |
Delisting: Proctol® ointment
As of May 2, 2026, Proctol® ointment, DIN 2247322, will become a PharmaCare non-benefit. Other therapeutic options (e.g., Proctodan-HC ointment Jampzinc-HC ointment, and Anodan-HC suppository) remain available and maintain regular benefit coverage.
| Drug name | dibucaine/esculin/framycetin/hydrocortisone 0.5/1/1/0.5% ointment (Proctol®) | ||
|---|---|---|---|
| Date | May 2, 2026 | ||
| Indication | Reduction of swelling, pain and inflammation associated with hemorrhoids and other rectal lesions | ||
| DIN | 02247322 | Strength & form | ointment |
Your Voice: Input needed for drug decisions
The knowledge and experience of patients, caregivers and patient groups is integral to B.C.'s drug review process. If you know someone who is taking one of the drugs below or who has a condition any of the drugs treat, please encourage them to visit www.gov.bc.ca/BCyourvoice.
Your Voice is now accepting input on the following drugs:
| Drug | Indication | Input window | ||
| roflumilast topical foam (Zoryve) | Plaque psoriasis of the scalp and body in patients 12 years of age and older | March 25 to April 21 at 11:59 pm | ||
| Iptacopan (Fabhalta) | The treatment of adult patients with complement 3 glomerulopathy (C3G) | March 25 to April 21 at 11:59 pm | ||

March 24 Special Release: BC PharmaCare expands coverage of diabetes-related supplies
As announced in the February 2026 PharmaCare Newsletter (PDF, 513KB), BC PharmaCare is expanding coverage of certain diabetes-related supplies, made possible by federal national pharmacare funding.
As of April 1, 2026, lancets, alcohol swabs and blood and urine ketone strips will be covered under Fair PharmaCare, Plan C (Income Assistance and Family Services), Plan F (Children in the At-Home Program), and Plan W (First Nations Health Benefits). Previously, these items were covered only by Plan W. These supplies will be covered up to a maximum price which includes an 8% markup, and annual quantity limits will apply. PharmaCare will not cover a dispensing fee for these supplies; these supplies are exempt from the Full Payment Policy.
As with blood glucose test strips, patients must receive training from a diabetes education centre (DEC) or a designated primary care network (PCN) to be eligible for coverage. Patients with an existing training certificate do not need to receive new training. The First Nations Health Authority (FNHA) will cover the first fill of eligible diabetes supplies for FNHA clients newly diagnosed with diabetes when needed. Contact FNHA at 1-855-550-5454 for coverage issues for FNHA clients.
PINs for diabetes supplies categories will remain the same as under Plan W, and will be posted on Diabetes product identification numbers (PINs).
| Product description | Annual quantity limit | Maximum price (incl. 8% markup) | PIN |
|---|---|---|---|
| Alcohol wipes/pads | 300 wipes | $0.0383 per unit | 11120001 |
| Blood ketone strips | 100 strips | $2.1060 per unit | 11120002 |
| Urine ketone strips | $0.1512 per unit | 11120003 | |
| Lancets for diabetic use | 400 units | $0.0756 per unit | 11120004 |
Alcohol swabs for non-diabetic use will continue to be covered under Plan W, reassigned to PIN 11120005. Pricing and annual quantity limits will align with the above table.
Resources
- February 2026 PharmaCare Newsletter (PDF, 513KB)
- PharmaCare Policy Manual, Section 5.10: Full Payment Policy
- Diabetes product identification numbers (PINs)
March 24 Special Release: No registration required for Plan NP coverage
B.C.'s National PharmaCare Plan (Plan NP) launched on March 1, 2026. Plan NP provides 100% coverage of select diabetes medications, menopausal hormone therapy (MHT), and contraceptives for any B.C. resident enrolled in the Medical Services Plan (MSP).
For B.C. residents enrolled in MSP, coverage is automatic. Clients do not need to be registered for Fair PharmaCare to receive Plan NP coverage.
Exceptional coverage under Plan NP is available to B.C. residents who have completed two-step enrollment and are in the wait period for MSP. For these clients, a pharmacist must apply for exceptional Plan NP.
Learn more about Plan NP at National Pharmacare (Plan NP).
Resources

About the PharmaCare Newsletter
The PharmaCare Newsletter is published on the first Wednesday of each month, with occasional mid-month releases. The PharmaCare Newsletter communicates drug listings, PharmaCare policy, PharmaNet procedures, and other pertinent information for PharmaCare providers and health care partners.
Information in previous newsletters is accurate as of the date it was published. Newsletters are not retroactively updated when policy, procedures or other information changes. Refer to the most recent mention of a topic for up-to-date information.
Search past newsletters on the Newsletter search page.
Subscribe
Enter your email address to subscribe to updates of this page.
 The PharmaCare Newsletter team works from the territory of the Lekwungen People, including the Songhees and Esquimalt Nations. Our gratitude extends to them, and all the Indigenous Peoples on whose territories and lands we build relationships.
The PharmaCare Newsletter team works from the territory of the Lekwungen People, including the Songhees and Esquimalt Nations. Our gratitude extends to them, and all the Indigenous Peoples on whose territories and lands we build relationships.
BC PharmaCare counts on pharmacy and device providers to practise cultural safety and humility.
To learn more, read Coming Together for Wellness, a series of articles by First Nations Health Authority (FNHA) and PharmaCare, and consider taking the online San’yas Indigenous Cultural Safety course.
Active advisories
spironolactone tablets; disopyramide capsules; olanzapine for injection; peginterferon alfa-2a (Pegasys®) injection
Visit Drug shortages for full list and details.