PAD Refills
June 2026

Prasterone (Intrarosa®) for the Genitourinary Syndrome of Menopause (GSM)
Download as a PDF
Read previous PAD Refills newsletters
The BC Provincial Academic Detailing (PAD) service’s topic Medications for menopause-associated vasomotor and genitourinary symptoms delivers evidence and practical drug information for the most commonly prescribed estrogen and progestogen products (oral, transdermal, vaginal).1 Visit www.bcpad.ca to book a session with an academic detailing pharmacist in your area.
We received this question during PAD sessions: Does vaginal prasterone offer an additional advantage on measures of sexual dysfunction compared to vaginal estrogen products accessible in the National Pharmacare formulary (Plan NP)?
| Basics1-4 |
|
|---|---|
| Comparisons5-6 |
|
What is the effect of vaginal prasterone ovules on sexual dysfunction in women with GSM?7
In one of the 12-week drug approval trials, the impact of prasterone on sexual dysfunction was assessed as a secondary outcome using the Female Sexual Function Index (FSFI) questionnaire. FSFI includes six domains: desire, arousal, lubrication, orgasm, satisfaction and pain. The total score ranges from 2 to 36 with higher scores indicating better or improved sexual function. Participants were postmenopausal women (mean age 60) with moderate to severe dyspareunia.
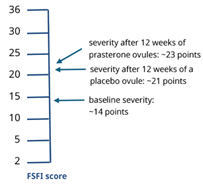
- From a baseline score of approximately 14 points, participants in the prasterone group achieved scores of approximately 23 after 12 weeks, compared to approximately 21 points in the placebo group
- In both groups, the average final score achieved remained within the definition of female sexual dysfunction (total score < 26.55)
- Prasterone has not been studied in women with a diagnosis of low sexual desire in the absence of moderate to severe genitourinary symptoms
*100% coverage (no cost to patient)
1BC PAD Service Medications for Menopause-Associated Vasomotor and Genitourinary Symptoms (May 2025); 2Health Canada Drug Product Database Intrarosa; 3Health Canada Clinical Information Intrarosa; 4Society of Obstetricians and Gynaecologists of Canada JOGC 2021 (PMID:34649681); 5Canada’s Drug Agency Intrarosa; 6Strandberg Maturitas (PMID:41903372); 7Labrie J Sex Med 2015 (PMID:26597311); 8Health Canada Drug Product Database; 9Cost: does not include mark-up or professional fee, calculated from McKesson Canada PharmaClik March 1, 2026; 10BC PharmaCare Formulary Plan NP menopausal hormone therapy
Please visit www.bcpad.ca to book a session with the academic detailing pharmacist in your area
2026
- May: Transdermal testosterone for hypoactive sexual desire disorder (HSDD) (PDF, 262KB)
- April: ADHD drug table update (PDF, 360KB)
- March: What is tibolone (Tibella)? The basis of Health Canada’s approval of tibolone for vasomotor symptoms associated with menopause. (PDF, 284KB)
- February: What is the role of venlafaxine (Effexor XR, generics) in the management of menopause-associated hot flashes? (PDF, 285KB)
- January: What is the role of Duavive in the management of menopause symptoms? (PDF, 284KB)
2025
- December: How effective is vaginal estrogen for the prevention of recurrent urinary tract infections associated with the genitourinary syndrome of menopause (GSM)? (PDF, 207KB)
- November: How does the weight loss effect of tirzepatide subcutaneous (Zepbound™) compare to semaglutide subcutaneous (Wegovy®) in people without type 2 diabetes? (PDF, 224KB)
- September: Do guidelines recommend initiating menopause hormone therapy (MHT) for the prevention or treatment of osteoporosis? (PDF, 178KB)
- June: Split, open, sprinkle: navigating ADHD medication formulations (PDF, 198KB)
- May: How does the evidence for tirzepatide subcutaneous (Mounjaro®) compare with semaglutide subcutaneous (Ozempic®) for type 2 diabetes? (PDF, 199KB)
- April: Antidepressant medication table (PDF, 186KB)
- March: Should dry powder inhalers (DPIs) be avoided in people with severe chronic obstructive pulmonary disease (COPD)? (PDF, 207KB)
- February: What do COPD guidelines recommend regarding inhaled corticosteriod (ICS) discontinuation in people with COPD? (PDF, 257KB)
- January: How does the weight loss effect of oral semaglutide (Rybelsus®) compare to subcutaneous semaglutide (Ozempic®, Wegovy®)? (PDF, 242KB)
2024
- December: Denosumab for osteoporosis: Two important updates (PDF, 263KB)
- November: What evidence exists for using lisdexamfetamine (Vyvanse®) in binge eating disorder in adults and how does this relate to weight loss? (PDF, 239KB)
- October: Which B.C. residents with mild-moderate COVID-19 are recommended to be treated with Paxlovid™ (nirmatrelvir/ritonavir)? (PDF, 258KB)
- September: Which factors should be considered when prescribing a bisphosphonate for frail or older adults living in long-term care? (PDF, 248KB)
- June: Why does a recent guideline recommend against the use of DPP4 inhibitors such as linagliptin (Trajenta®) in people with type 2 diabetes? (PDF, 241KB)
- May: What is the evidence for bupropion for ADHD in adults? (PDF, 239KB)
- April: COPD inhaled medication table update (PDF, 308KB) – updated in February 2025
- March: What is the role for orlistat (Xenical®) given the emergence of newer medications for weight loss in adults? (PDF, 270KB)
- February: What is romosozumab (Evenity™) and what is the evidence for it compared to bisphosphonates? (PDF, 241KB)
- January: What is the rationale for potentially holding GLP1 agonists, such as semaglutide (Ozempic®), preoperatively? (PDF, 253KB)
2023
- December: Is 300 mg per day the target dose of bupropion (Wellbutrin®) for major depressive disorder? (PDF, 293KB)
- November: When should serum calcium be checked in people initiating or taking denosumab (Prolia®) for fracture risk reduction? (PDF, 275KB)
- October: Should adults take 2000 IU of vitamin D daily to prevent fractures? (PDF, 273KB)
- September: How achievable is an HbA1c < 7% in people with type 2 diabetes when a second medication is added to metformin? (PDF, 304KB)
- June: Vyvanse and Dexadrine (PDF, 270KB)
- May: Switching to chlorthalidone compared to continuing hydrochlorothiazide: A new trial (PDF, 277KB)
- April: Zopiclone: dose, next-day impairment, tablet strengths (PDF, 261KB)
- February: Medications marketed in Canada for T2DM (PDF, 271KB)
- January: Melatonin supplementation in children with ADHD
2022
- December: SGLT2 inhibitors in patients with reduced kidney function (PDF, 285KB)
- November: Special Authority eForms (PDF, 321KB)
- October: Lipid-lowering medications (PDF, 303 KB)
- September: Increasing insulin glargine in people with type 2 diabetes (PDF, 253KB)
- June: The Why, Who, What, Where and How of nirmatrelvir/ritonavir (Paxlovid™) (PDF, 255KB)
- May: Osteoporosis Medication Table Update (PDF, 241KB)
- April: Can thiazides be continued in patients with chronic kidney disease when eGFR progresses below 30 mL/min/1.73m2? (PDF, 260KB)
- February: What do I need to know about sulfonylurea medications’ glucose lowering effect, dose and cost? (PDF, 247KB)
- January: What do I need to know about metformin’s glucose lowering effect, formulations, dose and cost? (PDF, 262KB)
2021
- December: Is quetiapine a safer alternative to zopiclone for insomnia? (PDF, 246KB)
- November: For people with type 2 diabetes, how does the oral daily formulation of semaglutide (Rybelsus®) compare with the subcutaneous weekly formulation (Ozempic®)? (PDF, 248KB)
- October: How does eszopiclone (Lunesta®) differ from zopiclone? (PDF, 242KB)
- September: Does vortioxetine (Trintellix®) improve cognitive functioning in people with major depressive disorder? (PDF, 242KB)
- June: What is lemborexant (Dayvigo) and how does it differ from other hypnotics for insomnia, such as zopiclone? (PDF, 247KB)
- May: Hypertension Medication Table Update (PDF, 251KB)
- April: Why and how should adverse drug reactions be reported? (PDF, 257KB)
- March: Is there a relationship between mirtazapine (Remeron®) dose and sedation? (PDF, 190.5KB)
- February: What is a prescribing cascade? The case of calcium channel blocker induced peripheral edema and loop diuretics (PDF, 188KB)
- January: Is venlafaxine (an SNRI antidepressant) more efficacious than SSRI antidepressants for generalized anxiety disorder? (PDF, 166KB)
2020
- December: Asthma Inhaled Medication Table Update (PDF, 216KB)
- November: COPD inhaled medication table update (PDF, 307KB) – updated in November 2024
Subscribe
Enter your email address to subscribe to updates of this page.
