Chronic Kidney Disease - Identification, Evaluation and Management of Adult Patients
Effective Date: October 30th, 2019
Recommendations and Topics
- Scope
- Key Recommendations
- Background
- Definition
- Etiology
- Risk Factors and Screening
- Diagnosis
- Referral Recommendations
- Management
- Lifestyle and Self-Management
- Complex Scenarios
- Abbreviations
- Resources
- Appendix A: Potential complications of CKD
- Appendix B: Interpretation of urine ACR values to assess albuminuria and proteinuria
- Appendix C: Recommended drug modifications in presence of CKD and in acute kidney injury (AKI)
Scope
This guideline provides recommendations for the investigation, evaluation, and management of adults at risk of or with known chronic kidney disease (CKD). These recommendations and treatment targets may not be appropriate in all cases because many patients with CKD are complex due to older age and comorbidities. Communication between primary care providers and specialists is strongly encouraged.
Key Recommendations
- Identify high-risk patient groups for evaluation of CKD: diabetes, hypertension, cardiovascular disease, family history, high risk ethnicity (Indigenous peoples, Pacific Islanders, African, Asian, and South Asian descent), history of acute kidney injury (AKI)
- Screen high-risk patients using eGFR and uACR. Confirm abnormal test results with a repeat measurement and obtain urinalysis
- Determine likely cause of kidney disease where possible. The cause of CKD has important implications for the risk of end stage renal disease (ESRD) and other complications
- The three dimensions of Cause, eGFR and Albuminuria (CGA) are all important in developing a management plan
- Prompt advice from local internists, local nephrologists or the RACE Line is available to assist in determining the need for and timing of referral
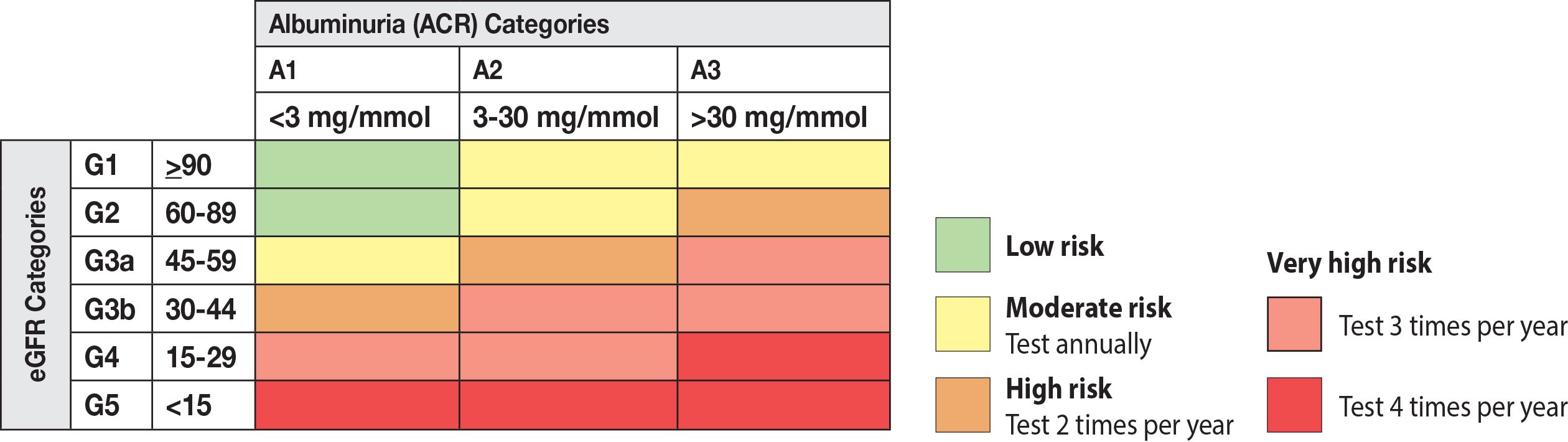
Figure 1. Prognosis and recommendations for frequency of monitoring based on eGFR and uAC*
* Adapted from ckdpathway.ca and Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney inter., Suppl. 2013;3:1–150
Background
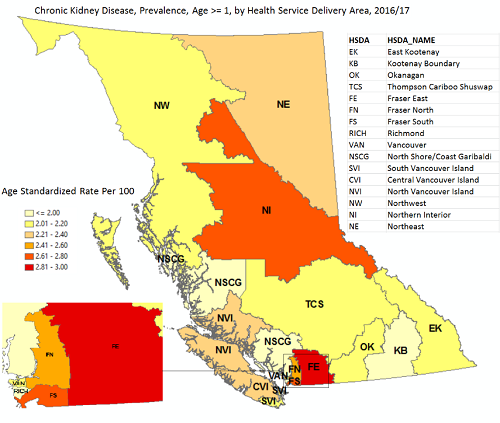
CKD is associated with other common chronic diseases such as diabetes, hypertension, and cardiovascular disease (CVD)1 and an estimated 1:10 British Columbians has some form of significant kidney disease2.
CKD markedly increases the risk of: cardiovascular disease, adverse drug reactions, acute kidney injury and prolonged hospital admissions4-10, 13-14. Patients with CKD have a risk of progression to end stage renal disease (ESRD), often requiring dialysis or kidney transplantation6, 9, 11-14. Most patients with chronic kidney disease die from other comorbidities before they progress to kidney failure. The outcome of many patients who go on to dialysis remains poor with 10 percent annual mortality; the overall 5 year survival rate is worse than that of most cancers3.
Evidence clearly indicates that control of hypertension and proteinuria can prevent or postpone kidney function decline4–12. This underscores the importance of early detection, evaluation and management of individuals with kidney disease.
Figure 2. CKD prevalence by health service delivery area, 2016/2017.
Definition
CKD is defined as an abnormality of kidney structure or function that is present for greater than 3 months13.
The two key parameters for classification are estimated glomerular filtration rate (eGFR) and urine albumin to creatinine ratio (uACR). eGFR is the best marker for kidney function and is calculated from creatinine. All labs in British Columbia (BC) automatically report eGFR when creatinine is ordered. Note: the recommended equations for eGFR may change over time and are estimates only. Refer to page 3 for more information.
For diagnostic purposes, other evidence of kidney damage includes: urine abnormalities such as hematuria; structural problems on imaging studies e.g. polycystic kidneys on ultrasound; histological findings on kidney biopsy.
Etiology
The two most common causes of CKD are hypertension and diabetes and they often co-exist13. Even if the cause seems obvious (e.g., diabetes), the possibility of a serious underlying primary renal disorder (e.g. glomerulonephritis) must be considered in patients with:
- Abnormal urinalysis, (e.g. proteinuria, hematuria, cellular casts). Note: hyaline casts are normal.
- Rapid sustained decline in kidney function (change in eGFR > 10-15%/year) despite remedy of reversible
precipitants (e.g. volume contraction, febrile illness, medications).
- Constitutional symptoms suggesting systemic illness.
- Sudden or severe onset of symptoms (e.g. edema unrelated to heart or liver disease).
Risk Factors and Screening
The following populations are at increased risk for CKD and should be screened:
- Diabetes
- Hypertension
- Cardiovascular disease (CVD)
- Prior acute kidney injury (AKI)
- Family history of kidney disease (e.g. parent or sibling)
- Specific high-risk ethnic groups: Indigenous peoples, Pacific Islanders, African Asian, and South Asian descent
Note: Older age alone is not a reason for screening.
At-risk populations should be screened every 1-2 years depending upon clinical circumstances (e.g., annually for individuals with diabetes) using:
- eGFR
- urinalysis (dipstick)*
- urine ACR
- review of risk factors
*Note: urine microscopy is not needed for screening.
Diagnosis
CKD cannot be diagnosed with one isolated abnormal measurement of eGFR or urine ACR.
Estimated GFR (eGFR) Values and Interpretation
- Values of > 60 mL/min per 1.73m2 without other markers of kidney disease (albuminuria, hematuria, structural abnormalities) do not indicate CKD
- Values of < 60 mL/min per 1.73m2 which are persistent (present for ≥ 3 months) are diagnostic of CKD
- A single isolated measurement < 60 mL/min does not satisfy diagnostic criteria for CKD, but could reflect reduced kidney function and requires confirmatory testing
- In patients with a new unexpected finding of reduced eGFR, the test should be repeated to establish stability or rapid deterioration
Caveats of eGFR
- eGFR is an estimated value that assumes a steady state of creatinine generation
- eGFR may be unreliable in extremes of muscle mass or with certain diets (e.g., very high or very low protein). Some medications can interfere with the excretion of creatinine (e.g., trimethoprim, fenofibrate14, 15
- In hospitalized patients or patients with AKI the fluctuations in creatinine make eGFR unreliable. In these circumstances, creatinine values should be used to guide management
- As a general rule, the eGFR can be used as a guide to outpatient drug dosing, even if the references use creatinine clearance (eCrCl). More caution is required for drugs with significant potential toxicity or a narrow therapeutic window (e.g. chemotherapy)
Urinalysis and ACR Values and Interpretation
- Significant abnormalities include persistent white blood cells or red blood cells in the absence of infection or instrumentation, and the presence of cellular casts
- Urine ACR (uACR) is the preferred method to screen for protein in the urine
- uACR quantifies albuminuria in a range which is not detected by dipstick urinalysis
- The term microalbumin has historically been used. Recent guidelines and consensus from laboratories have recommended abandoning this term and to quote the uACR value instead13
- uACR elevation (>3.0 mg/mmol) on serial testing is abnormal
- In patients without diabetes, no specific treatment is recommended for isolated uACR values between 3 and 30 mg/mmol. These individuals remain at risk of CKD progression and cardiovascular disease, therefore surveillance is warranted
- uACR may be unreliable in some patients due to acute illness, vigorous exercise, poorly controlled hypertension or poorly controlled blood glucose. Repeat testing should be done when in doubt
- Urine test abnormalities, even with persistent eGFR values ≥ 60 mL/min per 1.73m2 suggest kidney disease
- 24-hour urine collections are not necessary in most cases
- For equivalence of dipstick protein measurements, uACR and protein/creatinine ratios (PCR), see Appendix
Renal Imaging
- Renal ultrasound should be undertaken to assess structural abnormalities and aid in diagnosis. Renal ultrasound should be performed in the following cases:
- Obstructive urinary symptoms
- Unexplained microscopic or macroscopic hematuria
- Unclear etiology of CKD, especially in young patients
- Patients with suspicion of benign prostatic hypertrophy (BPH)
- Family history of structural renal disease (e.g. cystic kidney disease)
Staging of CKD
Risk staging of kidney disease is important for care planning and patient management. Risk is determined based on Cause, eGFR, and uACR, or CGA. Refer to Figure 1. Further details on risk determination are available on the Kidney Disease Improving Global Outcomes (KDIGO) CKD management guideline at kdigo.org/home/guidelines13.
Risk Calculators
The Kidney Failure Risk Equation (KFRE) is an equation designed to estimate probability of requiring dialysis within 2 or 5 years. The equation is derived from 4 variables and can be calculated using QxMD: qxmd.com. Risks of > 10 -20% indicate high risk (analogous to Framingham risk scores). The KFRE is a useful tool for prognostication, however its role has yet to be established in routine clinical practice.
Referral Recommendations
Indications for referral to specialist/nephrologist22 †
Very high risk kidney disease (urgent communication needed)
- Presence of active urine sediments (red blood cell casts or cellular casts ± protein), especially when associated with reduced eGFR
- AKI in absence of readily reversible cause (e.g. volume depletion, NSAIDs)
- Abrupt sustained fall in eGFR in a patient with known CKD
- eGFR < 15
- Nephrotic syndrome
High risk kidney disease (patient to be seen within a timely fashion)
- GFR < 30†
- Unexplained persistent uACR >30 mg/mmol (regardless of eGFR) e.g. in absence of diabetes or HTN
- Progressive CKD, with eGFR decline > 5 ml/min/year†
- Diabetes and evidence of CKD with eGFR <45, urine ACR >30
Low to moderate risk kidney disease (patients that could be seen within a longer time interval, e.g. within 6 mos)
- Persistent abnormalities of serum potassium
- Hereditary kidney disease
- CKD, eGFR 30-45
- CKD and difficult to treat hypertension
Recommended history and tests to include in referral package
- Comorbidities (especially cardiovascular)
- Medications
- Complete blood count (CBC)
- Electrolytes (Na, K, Cl, HCO3), calcium
- Creatinine/eGFR (include current value and any historical values available)
- Urinalysis (urine microscopy)
- Urine ACR
- If older than age 40: serum protein electrophoresis (SPEP), urine protein electrophoresis (UPEP)
- Renal ultrasound – not required prior to referral, but should be arranged with result sent to specialist when completed
Please use for referral
- Use Pathways to see the list of specialists in your region and their wait times
- Use the BC Renal website to locate nephrologist in your region: bcrenalagency.ca/kidney-services
- Real-time communication with local specialists (or RACE line if uncertain) can provide rapid advice for urgent cases and facilitate the most appropriate mechanism of referral
- Patients benefit from inclusion in multidisciplinary clinics for prevention, education and management, including by telehealth16–18
A urology consult is more appropriate than nephrology in the following scenarios
- Renal mass, enlarged prostate, obstruction, and large symptomatic or obstructing kidney stone
†Time to first consultation will vary based on individual circumstances. Discussion with your local nephrologist as to timing of referral for specific individuals is encouraged if in doubt.
Management
Refer to Figure 1 (page 1) for recommended follow-up intervals.
Customization of management plan
- Many patients with CKD have multiple comorbidities. In some situations, universal fixed targets may not be applicable and may even be harmful. Clinical judgment and individualized targets are recommended
- Communication with specialists is encouraged if appropriateness of any of the targets is in question
Table 1. Care objectives and targets for CKD
| Domain | Recommendations | Comments |
|---|---|---|
| Avoidance of acute kidney injury |
|
AKI is defined as an increase in creatinine by >26µmol/L or 1.5 times baseline. Transient insults to the kidney may result in a change in trajectory of stable kidney function. Risk of contrast-induced AKI is higher in people with volume depletion. |
| Medications |
|
Drug interactions are common in CKD and are avoidable. |
| Kidney function measurements |
|
|
| Blood pressure |
|
BP targets are continually changing. Use clinical judgment when interpreting BP targets. Consider comorbidities and prognosis. |
| CVD risk assessment |
|
After LDL reduction achieved, regular monitoring of lipids may not be necessary. |
| Diabetes |
|
HbA1C: ≤ 7.0% may not be appropriate for older frail patients. Risk/benefit of metformin can be challenging and requires discussion with a specialist. |
| Conditions associated with CKD |
|
Target hemoglobin is lower than the normal range for patients on ESA therapy (95- 115 g/L). Iron saturation <20% indicates iron depletion. Ferritin is not a reliable test in CKD patients and should not be ordered to assess iron deficiency. |
| Vaccinations |
|
Patients with very advanced CKD are less likely to seroconvert after hepatitis B immunization. Confirmation of immunity is required after vaccination, which may need to be repeated (after consultation with specialist) |
Lifestyle and Self-Management
Support patient self management with the means available in your community. Patients with CKD benefit from multidisciplinary renal teams who can support patients to make beneficial changes in diet, smoking and physical activity, and to understand medications, and the uncertainty and emotional distress that can be seen in those with chronic conditions. Denial, often associated with grief, is common in patients with chronic disease affecting a vital organ.
Encourage patients with CKD to consider advance care planning and their goals for future care, including for the end of life.
- Provincial advance care planning resources are available at http://www.bcrenal.ca/resource-gallery/Documents/Recommendations%20to%20Support%20Renal%20Patients%20at%20End%20of%20Life.pdf
- BC Guidelines on palliative care are available at https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/palliative-care
- BC Renal Agency information about end of life care is available at bcrenalagency.ca/healthcare-professionals/end-life-resources
Complex Scenarios
The following common complex scenarios benefit from discussion with a specialist:
- Whether to change the approach to a patient when eGFR goes down to 30 or lower
- Example: A 60 year old woman with type 2 diabetes mellitus has a change in eGFR from 32 to 29. Should her metformin be stopped?
- Deciding whether to stop diuretic therapy when the creatinine goes up or eGFR goes down
- Example: A 70 year old man with congestive heart failure requires escalation of diuretic therapy and experiences a deterioration in creatinine. Should his diuretics be discontinued?
- Difficult-to-treat hypertension
- Anticoagulation in patients with CKD
Abbreviations
- AKI: acute kidney injury
- CKD: chronic kidney disease
- CVD: cardiovascular disease
- eGFR: estimated glomerular filtration rate
- ESRD: end stage renal disease
- uACR: urine albumin to creatinine ratio
Resources
Diagnostic code: 585 (chronic renal failure)
Associated Documents
- Chronic Kidney Disease Flow Sheet
- List of Contributors
Appendices
- Appendix A: Potential complications of CKD
- Appendix B: Interpretation of urine ACR values to assess albuminuria and proteinuria
- Appendix C: Recommended drug modifications in presence of acute kidney injury (AKI)
Practitioner Resources
- BC Renal Agency - bcrenalagency.ca
Clinical resources for physicians, dieticians, pharmacists and information for patients.
- Chronic Kidney Disease Clinical Web/Mobile Tool: algorithm for CKD patient care
- RACE: Rapid Access to Consultative Expertise Program – raceconnect.ca
A telephone consultation line for select specialty services for physicians, nurse practitioners and medical residents. If the relevant specialty area is available through your local RACE line, please contact them first. Contact your local RACE line for the list of available specialty areas. Currently in BC, regions providing Nephrology services include Vancouver (VCH/PHC), Fraser and Northern. Regions that don’t currently have RACE support for Nephrology are Interior and South Island. If your local RACE line does not cover the relevant specialty service or there is no local RACE line in your area, please contact the Vancouver Coastal Health Region/Providence Health Care RACE line.- VCH Region/Providence Health Care: 604-696-2131 or 1-877-696-2131 (toll free)
- Northern RACE: 1-877-605-7223 (toll free)
- Fraser Valley RACE: raceapp.ca
- Pathways – PathwaysBC.ca
An online resource that allows GPs and nurse practitioners and their office staff to quickly access current and accurate referral information, including wait times and areas of expertise, for specialists and specialty clinics. In addition, Pathways makes available hundreds of patient and provider resources that are categorized and searchable.
- HealthLink BC: healthlinkbc.ca
HealthLink BC provides reliable non-emergency health information and advice to patients in BC. Information and advice on managing chronic kidney disease in several languages is available by telephone, website, a mobile app and a collection of print resources. Patients can speak to a health services navigator, registered dietitian, registered nurse, qualified exercise professional, or a pharmacist.
Patients may call 8-1-1 toll-free in B.C., or for the deaf and the hard of hearing, call 7-1-1.
- Island Health Community Virtual Care: Community Virtual Care provides support to people with a range of medical conditions. Registered nurses help you to manage your condition from the comfort of your home. All the tools needed are loaned to you at no cost.
- Health Data Coalition: hdcbc.ca
An online, physician-led data sharing platform that can assist you in assessing your own practice in areas such as chronic disease management or medication prescribing. HDC data can graphically represent patients in your practice with chronic kidney disease in a clear and simple fashion, allowing for reflection on practice and tracking improvements over time.
References
- Eckardt K-U, Coresh J, Devuyst O, Johnson RJ, Köttgen A, Levey AS, et al. Evolving importance of kidney disease: from subspecialty to global health burden. Lancet Lond Engl. 2013 Jul 13;382(9887):158–69.
- BC Renal Agency. Chances of having kidney disease estimated one in ten [Internet]. [cited 2014 Jun 12]. Available from: www.bcrenalagency.ca/news/chances-having-kidney-disease-estimated-one-10
- Canadian Institute for Health Information. CORR reports – treatment of end-stage organ failure in Canada 2001- 2010 (2011-12 Annual Report) [Internet]. [cited 2014 Jun 12]. Available from: //secure.cihi.ca/free_products/2011_CORR_Annua_Report_EN.pdf
- Trivedi HS, Pang MMH, Campbell A, Saab P. Slowing the progression of chronic renal failure: economic benefits and patients’ perspectives. Am J Kidney Dis Off J Natl Kidney Found. 2002 Apr;39(4):721–9.
- Snyder JJ, Collins AJ. Association of preventive health care with atherosclerotic heart disease and mortality in CKD. J Am Soc Nephrol JASN. 2009 Jul;20(7):1614–22.
- Diabetes Control and Complications Trial Research Group, Nathan DM, Genuth S, Lachin J, Cleary P, Crofford O, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993 30;329(14):977–86.
- Klahr S, Levey AS, Beck GJ, Caggiula AW, Hunsicker L, Kusek JW, et al. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of Diet in Renal Disease Study Group. N Engl J Med. 1994 Mar 31;330(13):877–84.
- Effect of intensive therapy on the development and progression of diabetic nephropathy in the Diabetes Control and Complications Trial. The Diabetes Control and Complications (DCCT) Research Group. Kidney Int. 1995 Jun;47(6):1703–20.
- Anderson S, Halter JB, Hazzard WR, Himmelfarb J, Horne FM, Kaysen GA, et al. Prediction, progression, and outcomes of chronic kidney disease in older adults. J Am Soc Nephrol JASN. 2009 Jun;20(6):1199–209.
- Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet Lond Engl. 2000 Jan 22;355(9200):253–9.
- Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001 Sep 20;345(12):861–9.
- ACE Inhibitors in Diabetic Nephropathy Trialist Group. Should all patients with type 1 diabetes mellitus and microalbuminuria receive angiotensin-converting enzyme inhibitors? A meta-analysis of individual patient data. Ann Intern Med. 2001 Mar 6;134(5):370–9.
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. Suppl. 2013(3):1–150.
- Paige NM, Nagami GT. The top 10 things nephrologists wish every primary care physician knew. Mayo Clin Proc. 2009 Feb;84(2):180–6.
- Shimada M, Dass B, Ejaz A. Assessment of elevated creatinine [Internet]. Available from: http://bestpractice.bmj.com/best-practice/monograph/935/overview/aetiology.html
- Goldstein M, Yassa T, Dacouris N, McFarlane P. Multidisciplinary predialysis care and morbidity and mortality of patients on dialysis. Am J Kidney Dis Off J Natl Kidney Found. 2004 Oct;44(4):706–14.
- Curtis BM, Ravani P, Malberti F, Kennett F, Taylor PA, Djurdjev O, et al. The short- and long-term impact of multi-disciplinary clinics in addition to standard nephrology care on patient outcomes. Nephrol Dial Transplant Off Publ Eur Dial Transpl Assoc - Eur Ren Assoc. 2005 Jan;20(1):147–54.
- Hemmelgarn BR, Manns BJ, Zhang J, Tonelli M, Klarenbach S, Walsh M, et al. Association between multidisciplinary care and survival for elderly patients with chronic kidney disease. J Am Soc Nephrol JASN. 2007 Mar;18(3):993–9.
- Owen RJ, Hiremath S, Myers A, Fraser-Hill M, Barrett BJ. Canadian Association of Radiologists consensus guidelines for the prevention of contrast-induced nephropathy: update 2012. Can Assoc Radiol J J Assoc Can Radiol. 2014 May;65(2):96–105.
- Komenda P, Zalunardo N, Burnett S, Love J, Buller C, Taylor P, et al. Conservative outpatient renoprotective protocol in patients with low GFR undergoing contrast angiography: a case series. Clin Exp Nephrol. 2007 Sep;11(3):209–13.
- Schachter ME, Romann A, Djurdev O, Levin A, Beaulieu M. The British Columbia Nephrologists' Access Study (BCNAS) - a prospective, health services interventional study to develop waiting time benchmarks and reduce wait times for out-patient nephrology consultations. BMC Nephrol 2013 Aug 29;14:182-2369-14-182.
- KCC Guidelines: http://www.bcrenalagency.ca/resource-gallery/Documents/Best%20Practices-%20Kidney%20Care%20Clinic_0.pdf
- Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJ, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, Cannon CP. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. New England Journal of Medicine. 2019 Jun 13;380(24):2295-306.
Appendix A: Potential complications of CKD
Potential Complications of CKD* (listed alphabetically)
- Acute Kidney Injury (e.g., dehydration, contrast dye, drugs)
- Anemia
- Hypertension
- Calcium absorption decreases
- Drug toxicity
- Dyslipidemia
- Heart failure/volume overload
- Hyperkalemia
- Hyperparathyroidism
- Hyperphosphatemia
- Hyperuricemia
- Left ventricular hypertrophy
- Malnutrition potential (late)
- Metabolic acidosis
* Listed complications are not specific to CKD but tend to occur with increasing frequency and are more directly attributable to more severe reduction in kidney function. If complications are noted at an early stage of CKD, investigation of alternative causes is recommended (e.g., profound anemia at eGFR of 55 mLl/min is likely not attributable to low kidney function alone). Kidney damage is defined as pathological abnormalities (e.g., kidney biopsy results) or markers of damage including abnormalities in blood or urine tests (e.g., protein/albumin in the urine, red blood cells, white blood cells or casts) or imaging studies.13
Appendix B: Interpretation of urine ACR values to assess albuminuria and proteinuria
|
Categories |
uACR (mg/mmol) |
Protein reagent strip |
PCR (mg/mmol) |
|---|---|---|---|
|
Normal to mildly increased |
< 3.0 |
Negative to trace |
< 15.0 |
|
Moderately increased |
3.0 – 30.0 |
Trace to + |
15.0 – 50.0 |
|
Severely increased |
> 30.0 |
+ or greater |
> 50.0 |
Appendix C: Recommended drug modifications in presence of CKD and in acute kidney injury (AKI)§
| Medication | Pathophysiology | Recommendation in CKD | Hold in AKI? |
|---|---|---|---|
|
NSAIDS |
Decreases renal perfusion, Interstitial nephritis |
Use with caution in CKD. |
Yes |
|
ACEI, ARB, or any medication containing those compounds |
Protective in proteinuric CKD, diabetes, and heart failure but can cause decreased renal perfusion and hyperkalemia |
ACEI or ARB should be held in hypovolemia, and if receiving IV contrast. |
Yes |
|
Potassium sparing diuretics (spironolactone, eplerenone, and amiloride) |
Volume depletion and hyperkalemia |
In CKD (other than in AKI), dose and frequent monitoring is essential if eGFR < 50 mL/min. |
Yes |
|
Metformin |
Increased risk of metformin associated lactic acidosis (MALA) |
Avoid if GFR <30 |
Yes |
|
SGLT-2 Inhibitors |
Protective in diabetic nephropathy and cardiovascular disease but can cause decreased renal perfusion in the setting of volume depletion |
Hold temporarily in the setting of volume depletion, or prior to administering contrast dye. |
Yes |
|
Diuretics: furosemide and thiazides |
Volume depletion and electrolyte abnormalities |
N/A |
Yes, unless volume overloaded |
|
Opioids (e.g. hydromorphone, fentanyl, methadone (for pain), oxycodone) |
Metabolites can accumulate |
Reduce dose in CKD For opioids that are considered safer in CKD, and opioids to avoid in CKD, consult: |
Consider dose reduction |
|
Pregabalin and gabapentin |
Accumulation |
Reduce dose and monitor for adverse effects In severe renal failure, dose should not exceed 300 mg gabapentin per day. |
Consider dose reduction |
|
Digoxin |
Hyperkalemia Accumulation with side effects (bradycardia, confusion) |
Reduce dose in CKD and monitor potassium and drug levels Consider alternative therapy in the setting of renal failure. |
Adjust dose |
|
Acyclovir |
Risk of crystal nephropathy Drug accumulation and side effects (risk of seizures/confusion) |
Encourage hydration. Dose adjust for GFR |
No. Ensure hydration. |
|
Statins |
Risk of rhabdomyolysis |
Consider dose reduction in CKD. Hold if rhabdomyolysis or unexplained / persistent muscle pain. |
No |
|
Phenytoin |
Risk of accumulation and toxicity |
Monitor levels and also adjust level for serum albumin |
No |
|
Lithium |
Accumulation and increased risk of side effects Risk of nephrogenic diabetes insipidus Risk of chronic interstitial nephritis |
Monitor lithium and electrolyte levels Encourage hydration Refer to a nephrologist if eGFR declines. |
No |
|
Hypoglycemics (sulfonylureas, insulin, meglitinides, thiazolidinediones) |
Accumulation can increase risk of hypoglycaemia |
Avoid long acting preparations in moderate-severe CKD. May require dose adjustments. |
No |
|
Trimethoprim and co-trimoxazole |
Risk of hyperkalemia Reduces tubular secretion of creatinine so leads to a rise in serum creatinine without renal injury |
Reduce dose when eGFR <30 mL/min, monitor GFR and potassium |
No |
|
Colchicine |
Risk of accumulation and serious toxicity (GI, CNS) |
Use lower dose and consider other agents (e.g. Steroids) |
No |
|
Proton pump inhibitors |
Risk of interstitial nephritis |
Clarify indication and consider other agent (e.g. H2 blocker) |
No |
For more information on dosing of DOACs in kidney disease please visit the 2018 European Heart Rhythm Association Guidelines (Figure 4). Available from academic.oup.com/eurheartj/article/39/16/1330/4942493
For more information on common prescribing questions for patients with CKD, consult:
http://www.bcrenal.ca/resource-gallery/Documents/Recommendations%20to%20Support%20Renal%20Patients%20at%20End%20of%20Life.pdf
Further information about drug dosage adjustments in chronic kidney disease is available from: rxfiles.ca/rxfiles/uploads/documents/ltc/HCPs/CKD/SDIS.Renal_newsletter.pdf
§Adapted from: Acute Kidney Injury – potentially problematic drugs and actions to take in primary care. “Think Kidneys” initiative by the UK Renal Registry in partnership with NHS England. Available from: thinkkidneys.nhs.uk/aki/wp-content/uploads/sites/2/2016/07/Primary-Care-Advice-for-medication-review-in-AKI-.pdf
This guideline is based on scientific evidence current as of the Effective Date.
This guideline was developed by the Guidelines and Protocols Advisory Committee, approved by the British Columbia Medical Association, and adopted by the Medical Services Commission.
For more information about how BC Guidelines are developed, refer to the GPAC Handbook available at BCGuidelines.ca: GPAC Handbook.
|
The principles of the Guidelines and Protocols Advisory Committee are to:
|
Disclaimer The Clinical Practice Guidelines (the "Guidelines") have been developed by the Guidelines and Protocols Advisory Committee on behalf of the Medical Services Commission. The Guidelines are intended to give an understanding of a clinical problem and outline one or more preferred approaches to the investigation and management of the problem. The Guidelines are not intended as a substitute for the advice or professional judgment of a health care professional, nor are they intended to be the only approach to the management of clinical problems. We cannot respond to patients or patient advocates requesting advice on issues related to medical conditions. If you need medical advice, please contact a health care professional.


 TOP
TOP