Follow-up of Colorectal Polyps or Cancer
Effective Date: January 16, 2013
Recommendations and Topics
Scope
This guideline provides follow-up recommendations for patients after curative resection of colorectal cancer (CRC) or polypectomy. These recommendations are intended to rationalize follow-up of the initial cancer and to prevent the development of additional colorectal cancer. They do not apply to patients with familial adenomatous polyposis (FAP), hereditary non-polyposis colon cancer (HNPCC) or inflammatory bowel disease. Recommendations for these patients and for the detection of colorectal neoplasms in asymptomatic patients are found in the guideline, BCGuidelines.ca - Colorectal Screening for Cancer Prevention in Asymptomatic Patients.
Key Recommendations
- Removal of adenomas can prevent CRCs.
- Individuals with colorectal adenomas or carcinoma are at high risk for recurrence.
- Colonoscopy is the key follow-up test to detect new primary cancers or adenomas.
- Patients followed by colonoscopy do not require fecal occult blood testing (FOBT).
- Early detection and treatment of CRC metastases may prolong survival.
Epidemiology
By definition all adenomatous polyps have dysplasia. Detection and removal of adenomas has been clearly demonstrated to reduce CRC mortality, and identification of cancer at an early stage markedly increases survival rates. Patients who have had CRC or advanced adenomas are at increased risk of recurrence and require close follow-up. The most important phase of follow-up is the first 2-3 years after the primary tumour resection as during this time the majority of recurrences will become apparent.1
The risk of an adenoma becoming malignant is greatest for "advanced" adenomas.
- tubular adenomas ≥ 1 cm,
- villous adenomas,
- adenomas with high grade dysplasia (HGD),
- sessile serrated polyps ≥ 1 cm,
- sessile serrated polyps with dysplasia, or
- traditional serrated adenoma.
Individuals with multiple (≥3) adenomas of any size are also at increased risk.2
It generally takes 5-10 years for a small adenoma to develop into a malignancy; cancer may be prevented by adenoma removal. 3
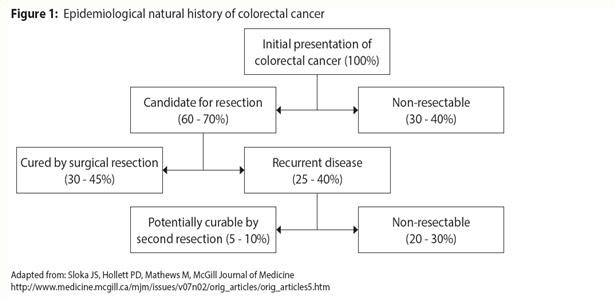
Follow-Up
Post-Polyectomy
The majority of CRCs arise from adenomas, the 'adenoma-carcinoma sequence'. Two major types of polyps are found in the colon and rectum: adenomas and hyperplastic polyps. Hyperplastic polyps are considered to have no malignant potential.
Table 1: Post-Colorectal Polypectomy Surveillance Recommendations4
|
Risk Group |
Surveillance Recommendations |
|
Patients with hyperplastic polyps |
Follow-up as average risk.* See BCGuidelines.ca - Colorectal Screening for Cancer Prevention in Asymptomatic Patients. |
|
Patients with 1 or 2 small (< 1 cm) tubular adenomas with only low-grade dysplasia |
Follow-up colonoscopy in 5 to 10 years. Timing within this interval should be based on other clinical factors (e.g., previous colonoscopy findings, family history, patient preferences, judgment of the physician). |
|
Patients with 1 or more sessile serrated polyps < 1 cm with no dysplasia |
Follow-up colonoscopy in 5 years. |
|
Patients with 3 to 10 tubular adenomas or any advanced adenomas (tubular adenomas ≥ 1 cm, villous adenomas, adenoma with HGD, sessile serrated polyps ≥ 1 cm, sessile serrated polyps with dysplasia, or traditional serrated adenoma) |
Follow-up colonoscopy in 3 years provided that adenomas are completely removed. If the follow-up colonoscopy is normal or shows only 1 or 2 small (< 1 cm) tubular adenomas with lowgrade dysplasia, the interval for the subsequent examination should be 5 years. |
|
Patients with sessile adenomas where complete removal is uncertain |
Follow-up colonoscopy within 6 months to verify complete removal. Once complete removal has been established, subsequent surveillance should be as for advanced adenomas. |
|
Patients suspected of having a hereditary colorectal cancer syndrome |
When the family history indicates HNPCC and FAP, colonoscopy every 1 to 2 years.** |
* FOBT is an appropriate follow-up modality for this group. FOBT should not be used until 10 years after the last colonoscopy for the hyperplastic polyp patient. All other risk groups above should not be followed with FOBT.
** Individuals with HNPCC or FAP should be referred to the Hereditary Cancer Program at the BC Cancer Agency for assessment, counseling and if appropriate, genetic testing.
Post-Cancer Resection
The goal of follow-up after resection is to identify recurrent disease or metastases and to detect subsequent adenomas. These recommendations are generally expert consensus-based. Patients with significant co-morbidities, very advanced age or limited 5 year life expectancy are not routinely offered surveillance.
Follow-up Visits with Family Physician
Focused history and physical examination are recommended every 3 to 6 months for 2 years, and then every 6 months for a total of 5 years.5,6 It is recommended that each follow-up visit include:
- History to elicit gastrointestinal and constitutional symptoms, including nutritional status.
- Physical examination with particular attention to the abdomen, liver and rectal evaluation (or perineal inspection and palpation in those patients who have had an abdominal perineal resection).
- Routine laboratory investigations, such as liver chemistry, in the absence of symptoms are not useful.
Controversies in Care
Aspirin (& other NSAIDS) has been shown to reduce the incidence of subsequent colorectal adenomas and cancer, but because of potential adverse effects it is currently not recommended.7
Tumour Markers
A carcinoembryonic antigen (CEA) test is recommended at diagnosis of CRC and repeated to monitor rising levels of CEA (at least doubling) which can indicate hepatic or pulmonary metastases. Eligible patients for surveillance with CEA are those with stage II or III tumors (i.e., tumour through the bowel wall or metastatic to locoregional lymph nodes). These patients are offered CEA every 3 months for the first 3 years and every 6 months during years 4 and 5. No CEA is required beyond 5 years.
Imaging and X-rays
Liver imaging, by ultrasound or CT scan (CT preferable),10,11 is recommended every 6 months for the first 3 years, then once per year for 2 more years.12 For those with advanced stage cancers or undergoing chemotherapy, follow the recommendations of the oncologist.5,13 Routine CT scanning is not recommended beyond 5 years.
There is little evidence to show a survival benefit for routine chest x-ray for post CRC resection patients.14 A chest CT scan is recommended for every 12 months for the first 3 years in cases of advanced cancer or rectal cancer.5,6
Colonoscopy
Patients with CRC should undergo a complete cancer and polyp clearing colonoscopy prior to or within 12 months of surgical resection of the colorectal tumour. A colonoscopy should follow at one year after resection or clearing colonoscopy.4,6 If the one year colonoscopy is normal, the next colonoscopy should be performed in 3 years; if those results are normal, the next colonoscopy should be performed in 5 years4,6 to look for another primary colorectal malignancy or adenomatous polyps. After the one year colonoscopy, the intervals between subsequent colonoscopies may be shortened if there is evidence of HNPCC or if adenoma findings warrant earlier colonoscopy.
Performance of FOBT is unnecessary in patients undergoing colonoscopic surveillance.9
Surveillance After 5 Years
Continued surveillance is recommended with a colonoscopy conducted every 5 years. There is no place for FOBT in this population.
Resources
References
- Jeffery M, Hickey BE, Hider PN. Follow-up strategies for patients treated for non-metastatic colorectal cancer. Cochrane Database of Systematic Reviews 2007, Issue 1.
- American Society of Gastrointestinal Endoscopy. ASGE guideline: colorectal cancer screening and surveillance. Gastrointest Endosc. 2006;63:546-57.
- Zauber AG, Winawer SJ, O'Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. NEJM. 2012;366:687-696.
- Brooks DD, Winawer SJ, Rex DK, et al. Colonoscopy surveillance after polypectomy and colorectal cancer resection. Am Fam Physician. 2008;77(7):995-1002.
- Desch CE, Benson AB, Somerfield MR, et al. Colorectal cancer surveillance: 2005 update of an American Society of Clinical Oncology practice guideline. J Clin Oncol. 2005;23:8512-8519.
- National Comprehensive Cancer Network®. NCCN Guidelines™ Version 3.2012: Colon Cancer. c2012 [updated 2012 Jan 17; cited 2012 Apr 13]. Available from http://www.nccn.org/clinical.asp
- Din FV, Theodoratou E, Farrington SM, et al. Effect of aspirin and NSAIDs on risk and survival from colorectal cancer. Gut 2010;59:1670-1679.
- Locker GY, Hamilton S, Harris J, et al. ASCO 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol. 2006;24(33):5313-27.
- Winawer SJ, Zauber AG, Fletcher RH, et al. Guidelines for Colonoscopy surveillance after polypectomy: A concensus update by the US Multi-Society Task Force on Colorectal Cancer and the American Cancer Society. CA Cancer J Clin. 2006;56:143-159.
- Kinkel K, Lu Y, Both M, et al. Detection of hepatic metastases from cancers of the gastrointestinal tract by using noninvasive imaging methods (US, CT, MRI, PET): a meta-analysis. Radiology 2002;224:748-756.
- Miles K, Burkill G. Colorectal cancer: Imaging surveillance following resection of primary tumour. Cancer Imaging 2007;7:S143-S149.
- Figueredo A, Rumble RB, Maroun J, et al. Follow-up of patients with curatively resected colorectal cancer: A practice guideline. BMC Cancer. 2003;3:26.
- Pfister DG, Benson AB, and Somerfield MR. Surveillance Strategies after curative treatment of colorectal cancer. N Engl J Med. 2004;350:2375-2382.
- Gan S, Wilson K, Hollington P. Surveillance of patients following surgery with curative intent for colorectal cancer. World J Gastroenterol. 2007;13(28):3816-3823.
Resources
- BC Cancer Agency, Follow-up Program after Colorectal Cancer Treatments,www.bccancer.bc.ca/books/Documents/Gastrointestinal/PatientBrochureFollowupProgramafterColorectalCance.pdf
- HealthlinkBC - Health information, translation services and dieticians, www.healthlinkbc.ca
- Canadian Cancer Society, www.cancer.ca
- Colorectal Cancer Association of Canada, www.colorectal-cancer.ca
- Colon Cancer Canada, www.coloncancercanada.ca
Associated Documents
- Patient Guide (PDF, 504KB)
- Guideline Summary (PDF, 194KB)
- BCGuidelines.ca – Colorectal Screening for Cancer Prevention in Asymptomatic Patients (2013)
This guideline is based on scientific evidence current as of the Effective Date.
This guideline was developed by the Guidelines and Protocols Advisory Committee, approved by the British Columbia Medical Association, and adopted by the Medical Services Commission.
|
The principles of the Guidelines and Protocols Advisory Committee are to:
|
Disclaimer The Clinical Practice Guidelines (the "Guidelines") have been developed by the Guidelines and Protocols Advisory Committee on behalf of the Medical Services Commission. The Guidelines are intended to give an understanding of a clinical problem and outline one or more preferred approaches to the investigation and management of the problem. The Guidelines are not intended as a substitute for the advice or professional judgment of a health care professional, nor are they intended to be the only approach to the management of clinical problems. We cannot respond to patients or patient advocates requesting advice on issues related to medical conditions. If you need medical advice, please contact a health care professional.


 TOP
TOP