Forest fertilization guidebook
The Forest and Range Practices Act and its regulations took effect on Jan. 31, 2004. It replaced the Forest Practices Code of British Columbia Act and regulations. Forest Practices Code guidance is presented for information purposes only.
The guidance offered here is still relevant with regard to field practice.
Introduction
Purpose
This guidebook has been prepared to assist forestry practitioners in meeting the requirements of the Forest Practices Code with respect to the safe and efficient, broadcast application of fertilizer.
Specific Forest Practices Regulations and Standards for forest fertilization are listed with relevant guidelines enclosed in text boxes throughout the guidebook. Guidelines cover the selection of suitable stands, fertilization methods, and monitoring procedures required to implement a forest fertilization operation.
Response to fertilization
Fertilization is a silvicultural treatment that can be effectively used to increase the merchantable yield and value of established forests. By adding nutrients that are limited on a site, fertilizers can improve the growth of individual stands. However, with knowledge of the timber supply profile and the timing and magnitude of wood supply needs, fertilization can also be strategically used to accelerate the development of specific age classes and timber types. This is done to facilitate an even supply of wood at the forest level.
The response of a forest stand to fertilization is best considered as a reduction in rotation length. Fertilization accelerates the rate of stand development. A fertilized crop will therefore generally not differ significantly from a non-fertilized crop grown over a longer rotation.
Although fertilizers are normally applied to accelerate stand development, an alternative objective is the rehabilitation of disturbed sites. Secondary roads and landings may experience excessive soil disturbance. Rehabilitation of these areas may require the application of fertilizer to promote a cover crop and reduce erosion. Procedures for rehabilitating sites can be found in Soil Disturbance Rehabilitation Guidebook.
Fertilizer can also be applied at the time of planting. This is done on sites where:
- the growth of seedlings is inhibited by inadequate nutrient availability
- rapid growth is required to reduce competition with other vegetation
- rapid early growth is desired to meet forest level objectives
Fertilization at planting is normally done with a slow release fertilizer in or near the planting hole. The methods and procedures discussed in this guidebook describe broadcast fertilizer applications used to increase tree growth.
Authority
Operational Planning Regulation:
Fertilizing done under a silviculture prescription
A person who is required to establish a free growing stand on an area under prescription must specify a fertilization treatment if it is required to achieve a healthy free growing stand within the latest free growing date specified in the prescription, unless such a treatment is in a community watershed and is precluded by the objectives of the community watershed.
The Operational Planning Regulation gives five points that a person preparing a stand management prescription must do to meet the required scope of a stand management prescription.
Scope of stand mangement prescriptions
(a) consider the result of any free growing survey carried out on the area,
(b) ensure that the prescription is adequate to achieve the future stand condition specified in the prescription,
(c) ensure that the prescription is adequate to achieve continued growth of species that are ecologically suited for the area,
(d) ensure that the prescription facilitates the protection of the soil on the area, and
(e) describe any actions required to achieve known landscape level objectives for stand structure and species composition.
Stand management must:
- contains a reasonable evaluation of any forest health factors that exist currently in the area or that may reasonably be expected to occur in the area before the harvest of the next crop and describes the actions that are proposed to deal with those factors,
- sets out the target future stand condition and proposed treatments, including spacing, pruning and fertilizing and the objectives of those treatments,
- sets out the stand tending treatment requirements
Silviculture Practices Regulation:
Stand management
Fertilization mandatory in some circumstances: A person who is required to establish a free growing stand on an area under a silviculture prescription must carry out a fertilization treatment on the area before the end of the free growing assessment period, if the district manager is of the opinion that the treatment is necessary to achieve a healthy free growing stand within the free growing assessment period specified in the prescription.
Use of fertilizers
A person who uses fertilizer to
(a) establish a free growing stand on an area under a silviculture prescription, or
(b) carry out a stand management prescription
must store, handle and apply the fertilizer in a manner that protects forest resources.
Broadcast fertilization in community watersheds
A holder of a silviculture prescription or stand management prescription who broadcasts fertilizer on an area under the prescription in a community watershed must ensure that
(a) the fertilizer is not applied
(i) closer than 100 m upslope of a water intake, or
(ii) within 10 m of a perennial stream that is observable from the air, at the height the fertilizer will be applied, unless otherwise authorized by both the district manager and the Minister of Health, and
(b) the application of nitrogen fertilizer does not cause
(i) nitrate nitrogen levels in a stream to exceed 10 ppm measured immediately below the area where the fertilizer is applied,
(ii) chlorophyll levels in a stream to exceed
(A) 2 micrograms/litre in a lake, or
(B) 5 milligrams/square metre in a stream, or
(iii) water quality to fall below any water quality objectives or criteria approved by the Minister of Environment, Lands and Parks.
Reports
Obligation of major license holder under stand management prescription
(1) A holder of a major licence who carries out a silviculture treatment specified in a stand management prescription approved under section 24(2) of the Act must prepare a report that
(a) contains a summary of the silviculture treatments performed on the free growing stand on the area during the period beginning 2 weeks before the last report and ending 2 weeks before the current report’s submission date under subsection (2), and
(b) specifies whether the silviculture treatment was carried out to fulfill the requirements of a management plan or an agreement under the Forest Act.
(2) A person required to prepare a report under subsection (1) must ensure that the report
(a) is signed and sealed by a professional forester, and
(b) is submitted each year to the district manager on April 15, July 31, October 31 and January 15.
(3) The chief forester may specify the form and content of a report under subsection (1).
Fertilization required by legislation
A silviculture prescription must contain a requirement for fertilization if the district manager determines that it is necessary to obtain a free growing stand. This requirement will only be removed if the holder of the silviculture prescription can demonstrate that fertilization is unlikely to significantly improve stand vigor. This demonstration would be done using evaluation of site and stand characteristics and, documentation of stand nutrient status by foliar analysis and fertilizer screening trials.
Fertilization should be a requirement of a silviculture prescription if necessary for the stand to meet required growth levels. Fertilizer should not be required if its application will cause other site or crop tree problems.
Guideline
The district manager can determine fertilization to be a free growing requirement.
Stand selection
Forest level planning
Forest level plans may require an increase in forest production in order to meet timber supply objectives. A large-scale forest fertilization program may help meet these objectives. In most cases the yield and value benefits of fertilization at the stand level are maximized by combining nutrient additions with a spacing treatment. Spacing will concentrate growth increases on fewer stems and provide growing space for the crowns of fertilized trees. Plan spacing treatments to first meet forest or landscape level objectives.
Stand level planning
The stand level objectives for a fertilization treatment are chosen considering a stand’s site conditions, health, biodiversity and potential for integrated resource management. Stand level objectives should be consistent with forest level objectives. The costs incurred to achieve the necessary stand level objectives should be kept at a minimum.
With the decision to fertilize comes the need to evaluate candidate stands for suitability and priority.
Guideline
Evaluate candidate stands according to biological factors. Those stands that are biologically acceptable should then be checked for operational feasibility to ensure they can indeed be treated and are suitable for treatment.
The following section explains in detail the major factors to be considered. These factors, which should be considered in the development of stand mangement prescriptions and stand selection, are summarized in Appendix 1. When assigning a priority to a particular stand, consider as many factors as possible. These ranking criteria were developed for stands when the main objective of the fertilization is an increase in net merchantable volume growth, or earlier harvest. This ranking criteria must be modified if the objectives of the treatment are different.
Biological principles
Trees respond to added nutrients by increasing the rate of photosynthesis per unit of foliage area (i.e., photosynthetic efficiency) and by increasing photosynthetic surface area through the production of more foliage and expansion of live crowns. These response mechanisms will apply regardless of the treatment objectives.
Increased bole wood production during the first year after fertilization is primarily due to increased photosynthetic efficiency caused by higher foliar nitrogen concentration. However, foliar nitrogen levels of fertilized trees generally return to pre-fertilization levels after about three years. It is the increased foliage mass caused by increased needle size, number of needles per shoot, and number of shoots that results in enhanced bole wood production over the majority of the response period (five or more years). This is why it is critical that crop trees have room for crown expansion following fertilization. If not, the growth response to fertilization will be limited to the short-lived increase in photosynthetic efficiency, rather than the prolonged response due to increased foliage mass.
The growth response to fertilization is largely dependent on the amount of the added nitrogen that is taken up by trees during the short period following treatment. In most forest soils, urea fertilizer is quickly converted to ammonium (NH4+) nitrogen, which is readily taken up by trees and other vegetation. However, the recovery of added nitrogen in crop trees is generally quite low, ranging from less than 10% to approximately 30%. Most of the added nitrogen is rapidly immobilized in soil microbial biomass and organic matter. The immobilized nitrogen is largely unavailable for tree uptake and is generally mineralized too slowly to have much practical value in improving the growth of crop trees.
Under certain conditions, significant losses of added nitrogen can occur from gaseous losses of ammonia (NH3). Volatilization losses will increase with high air temperature, wind speed, and soil pH. Volatilization can be minimized by timing nitrogen applications to coincide with cool (<10°C), calm weather with a high probability of rain in the next 24 hours.
Biological factors
Biological factors to be considered during the stand treatment ranking are listed under the following eight headings.
Species
Response to fertilization is discussed in terms of coastal and interior species:
-Coastal species
For coastal areas, Douglas-fir has shown consistent and significant growth response to nitrogen fertilization. Reliable methods exist for predicting response of Douglas-fir to fertilization. Stands with >80% Douglas-fir component should be assigned the highest priority. Fertilizing Douglas-fir outside of its normal ecological range (see Establishment to Free Growing Guidebook) is not recommended, as those stands may have greater susceptibility to snow and frost damage.
Western hemlock has shown erratic response to fertilization. More consistent response to N + P applications has been shown in young stands growing on substrates rich in decaying wood in the CWHvh subzone and salal-prone sites at low elevations in the CWHvm subzone. On other sites, hemlock stands should only be considered for research or operational trials.
Limited local experience indicates that Sitka spruce stands, particularly those in chlorotic condition, respond to fertilizing. Fertilization of Sitka spruce may increase the incidence of leader attack by the spruce weevil (Pinus strobi). Until research or operational trial results better document positive results, extensive fertilizing should be delayed and a low priority assigned to this species. Screening trials are recommended prior to treatment.
For commercial tree species other than Douglas-fir, Sitka spruce, and western hemlock, as discussed above, fertilizer application should be restricted to research installations and operational trials.
For coast–interior transition areas, little information is available about growth responses of trees to fertilizer. Only consider Douglas-fir stands for inclusion in large-scale fertilizer programs. Also note that this recommendation is based entirely on extrapolation of coastal experience. To ensure soil moisture does not seriously limit growth responses, do not fertilize on sites with more than three months of growing season water deficit (e.g., avoid very dry and moderately dry soil moisture regimes).
Table 1 (right). Species-specific site characteristics that may be useful indicators of nutrient deficiencies. (From: Soo TSA Forest Fertilization 10 Year Plan, R.E. Carter, E.R.G. McWilliams.)
-Interior species
Research throughout the B.C. interior has shown that lodgepole pine is consistently deficient in nitrogen. Nitrogen additions often have a substantial positive effect on tree and stand growth. On sites with marginal sulphur status (<60 ppm foliar sulphate – S), fertilizer nitrogen response may be improved when sulphur is applied with the nitrogen. Induced boron deficiencies following nitrogen fertilization have also been documented. Boron deficiency causes top dieback which can have a large negative impact on stem value. The addition of a small amount of B in the operational fertilizer mix is generally effective in preventing damage. However, it is probably most cost effective to avoid fertilizing stands where pre-fertilization foliar analysis indicates a potential B problem (i.e., pre-fertilization foliar B <10 ppm).
There is currently limited fertilization response information for Douglas-fir in the B.C. interior. However, results from fertilizer trials in the Intermountain region of the United States indicate that N fertilization has produced consistent growth response over a broad range of site and stand conditions. Douglas-fir stands in northern Idaho have responded very well to N additions. In many respects, these stands are similar to Douglas-fir growing in the Interior Cedar-Hemlock (ICH) Biogeoclimatic Zone in south-central B.C. However, extensive fertilization should not be undertaken until research or operational trial results document positive results for wet-belt Douglas-fir in southern B.C.
Other species, including dry-belt Douglas-fir, black spruce, white spruce, western redcedar, western hemlock, and western larch, are recommended for trials only until more response information is available.
Age and size
Discussion of the influence of age and size on fertilizer response is broken into two sections:
-Coastal
For Douglas-fir there is no clear relationship between stand age and the percentage response in periodic increment following fertilization. Factors such as crown vigor and room for crown expansion are apparently more important than age in predicting the response. Since the volume increment of a stand tends to decline with age, the absolute increases in volume from fertilization will also decline with age. However, from a financial viewpoint, older immature stands might be the most desirable investment. The costs of fertilizing will be compounded for only a few years before harvest. Therefore, provided that live crowns are of favorable size and vigor, and there is room for crown expansion, preference should be given to fertilizing older stands rather than younger ones. Stands recently commercially thinned, with favorable stand structure are good candidates for fertilizing in these older age classes. Given these conditions and the absence of priority ratings based on the need to mitigate specific age class distribution gaps, the highest priority should be assigned to stands 10–15 years from expected rotation end, followed in order of preference, by stands 16–30 years, and 31 or more years from rotation end.
Generally, very young Douglas-fir (15–20 years) should not be fertilized until trees are at least 2 m taller than the ground vegetation. This height advantage is necessary so that competing vegetation will not overtake the crop trees. An exception is when early fertilization is necessary to meet free growing or green-up requirements.
-Interior
As with coastal species, preference should be given to fertilizing older stands in the interior, provided that live crowns are of favorable size and vigor, there is room for crown expansion, and that other forest level objectives do not take priority. Older, spaced stands and unspaced stands shown to have appropriate structures for fertilization (i.e., naturally occurring lower density or a suitable number of well-spaced dominants) or good potential for spacing or commercial thinning (i.e., healthy, vigorous crowns) may be assigned a high priority. However, the yield implications should be carefully considered before these stands are spaced or commercially thinned to provide fertilization opportunities.
Unfortunately, many older, unmanaged interior stands (e.g., age class 3 or 4 lodgepole pine) exhibit poor fertilization response potential. These stands often have unfavorable stand structure and low potential for response to spacing or commercial thinning. As such, combined fertilization and thinning treatments in older, unmanaged stands with questionable structure is recommended for operational trials only until their responsiveness to treatment is documented. Fertilization should probably be delayed 3–5 years after spacing or commercial thinning in these stands in order to allow the remaining crop trees to adjust to the new environment and develop sufficient new foliage to utilize the added nutrients.
Because of the unfavorable structure of many older stands in the B.C. interior, the highest fertilization priority is generally assigned to 15- to 40-year-old stands. As with coastal Douglas-fir, stands 15–20 years old should not be fertilized unless trees are at least 2 m taller than competing vegetation. Very young spaced stands or plantations (under 15 years) may exhibit a large relative response to fertilizer additions but a small absolute stem volume response due to their small stem diameter. The site occupancy of such stands may also be too low to efficiently utilize the applied fertilizer.
Stand density
The magnitude of fertilizer response is related to the space available for crown expansion. Stands selected for fertilization should have well-spaced dominant and codominant trees. This will often mean that stands will be spaced before fertilization. It is the decision to space a stand that must take precedence over fertilization. Therefore, choose stands for spacing whose treatment will meet higher level objectives. These stands can then be candidate stands for fertilization.
Priority for fertilization:
- Pruned stands with favorable spatial distribution.
- Stands with favorable spatial distribution.
- Stands scheduled to be spaced within a year of the fertilizer treatment.
Fertilizer treatment of recently spaced stands can reduce spacing shock. In most cases the best time to fertilize is at the time of spacing. Fertilization response data for lodgepole pine indicate that relative and absolute volume responses are generally larger for stands fertilized at the time of spacing compared to fertilization 2–3 years after spacing. However, fertilization should be delayed if one or more of the following factors exist:
a. The live crown of the remaining crop trees is of insufficient size (e.g., <30%) to utilize the added nutrients.
b. The height/diameter breast height (dbh) ratio of remaining crop trees is large enough to put trees at risk of toppling and breakage. Fertilized trees produce substantially more foliage the year following treatment, and the larger crowns increase the susceptibility of snow and wind damage. For Douglas-fir, it is recommended that the height/dbh ratio should be less than 85. No conifer stands with a height/dbh ratio greater than 100 should be fertilized. The same criteria apply for spaced lodgepole pine. Lodgepole pine stands with pre-spacing densities >10 000 have been shown to be especially susceptible to snow press where spacing and fertilization have been undertaken simultaneously. In these stands, risks can be substantially reduced by delaying fertilization for 2–3 years after spacing.
c. Fertilization should be delayed 1–3 years in situations where there is a heavy cover of fine thinning slash. Decomposition of slash can act as a “green manure,” thereby providing a short-term increase in nutrient availability. A heavy cover of slash can also prevent fertilizer prills from reaching the soil, thus increasing risks of N volatilization losses.
d. In older age class 3 and 4 stands, fertilization should be delayed for 3–5 years after spacing or commercial thinning unless pre-treatment densities were below 4000 stems per hectare and there is no reason to anticipate any wind throw losses.
Guideline
Fertilizing too soon after spacing may increase the risks of wind or snow damage. Delay fertilizing for two or more years after spacing when:
- live crowns are <30% (unless the healthy live crown is >6–8 m in length)
- height/dbh ratio >100
- pre-spacing densities of lodgepole pine are >10 000 sph
- there is a heavy layer of fine slash after spacing
- the stand is a older age 3 or 4 lodgepole pine stand with pre spacing densities >4000 sph
Soil moisture and nutrient regimes
Moisture and nutrient regimes of forest soils affect tree responses to fertilizing and must therefore be carefully considered. The most reliable information on soil nutrient availability is obtained from foliar analysis. While evidence of chlorotic tree foliage can indicate soil infertility, caution should be used in making interpretations based on these indications (see Crown condition). Lastly, the plant community often reflects soil moisture and nutrient regimes. Use regional guides to relate plant associations to soil moisture and nutrient conditions.
Soil moisture regime
For coastal Douglas-fir, the largest and most consistent responses are found on slightly dry to fresh sites (e.g., submesic and mesic). In the B.C. interior, sites that are slightly dry to fresh should receive the highest priority (e.g., submesic and mesic), with lesser priority assigned to drier (subxeric) and wetter (subhygric and hygric) sites. Because soil moisture, either by deficiency or excess, may exert the primary limitation on tree growth, fertilization of stands with very dry (xeric and very xeric) or very wet (subhydric) conditions should be avoided.
Nutrient regime
Although benefits from fertilizing occur most consistently on infertile sites, very infertile sites (site quality is low) are unsuitable for treatment because natural growth rates are too slow. Avoid very rich sites also, since the soil probably contains adequate nutrients. Stands on poor and medium regimes should respond best, and therefore will be given the highest priority. Moisture and nutrient regimes are two of the main factors that determine site quality as discussed in the following section.
Guideline
- Do not treat stands on very dry or very wet sites where water is the limiting factor for tree growth.
- Do not treat stands on very poor or very rich sites where nitrogen is not the limiting factor for tree growth.
Site quality
Site quality considerations vary between coastal and interior sites.
-Coastal
Research data in coastal Douglas-fir stands indicate that the magnitude of growth response to fertilizer application is greater on poorer sites than on good sites. Based on the current Inventory Branch (Ministry of Forests) classification, preference should therefore be given to fertilizing poor and medium sites. Response on good sites will be small relative to the size of the trees. However, the absolute response on good sites (periodic increment) can be large compared to the response on medium or poor sites. Fertilization on good sites should be considered when a large absolute response is predicted (e.g., on sites having a moist soil moisture regime and medium soil nutrient regime). Fertilizer should not be applied on low sites, since they are unlikely to ever become economically productive.
-Interior
Preference should be given to fertilizing medium sites (e.g., submesic to mesic soil moisture regimes and poor to medium soil nutrient regimes) in the B.C. interior. A lower priority should be assigned to good sites until such time that additional research information is available. Fertilization on poor sites will have low priority since site productivity is probably influenced strongly by inadequate soil moisture or extreme climate. Even where relative growth responses are favorable, the absolute volume gains on these lower productivity sites may be too small to make fertilization profitable. Fertilizer should not be applied on low sites.
Crown condition
The size and condition of live crowns provide an indication of the nutrient status and productive potential of stands. Considerations related to crown size and foliar characteristics are described below.
Size
In unspaced and recently spaced stands, evidence of many trees with short, narrow crowns suggests competitive stress is, or has been, strong. Application of fertilizer to stands in this condition will enhance crown expansion by stimulating growth of branches and foliage. Assign a priority to the treatment of these stands provided they meet the criteria described in the guideline in the section on stand density.
Foliage color
Small, yellowish, and sparse foliage throughout the stand may indicate that one or more soil nutrients are deficient. In this situation fertilization may achieve a substantial growth response. However a chlorotic appearance may also be caused by drought or pathological conditions. In the absence of chemical analysis of soils or foliage, the interpretation of visual symptoms requires expertise and local knowledge. Look for other symptoms that may identify insect, disease or animal damage. Also consider soil conditions, ground vegetation, and rainfall patterns to infer drought.
If foliar appearance in a stand can be reliably interpreted to indicate nutrient deficiency and sufficient response is anticipated to make the treatment economical, the site can be assigned a high priority for fertilizing. However, an absence of visual symptoms does not preclude the possibility of growth limiting nutrient deficiencies.
Nutrient diagnosis
-Foliar analysis
Foliar analysis is a useful method of indicating possible nutrient deficiencies. Foliar analysis should only be undertaken after a candidate stand meets all other biological, operational, and wood supply criteria. Because of the cost of sampling, foliar analysis should only be done on sites which have the potential for a return on investment. It is also important to assess soil and site conditions in conjunction with foliar analysis in order to facilitate the interpretation of analytical results.
Tables 1 and 2 in Appendix 2, show interpretations of macro and micronutrient foliar concentrations. Consult a tree nutrition specialist when interpreting results of foliar analysis.
Appendix 2 outlines the foliar sampling procedure.
Adequate time must be scheduled for foliar analysis. Labs routinely take 4–6 weeks to analyze samples.
On the coast, sampling of candidate Douglas-fir stands is not normally required. However, foliar sampling should be carried out if local experience suggests that multiple nutritional problems are likely. Candidate hemlock and spruce should be analyzed to ensure that they are N deficient and that other nutrients (e.g., phosphorus) will not limit response.
Based on the results of the analysis, stands that are nitrogen (N) deficient only, with no potential N-induced deficiencies, should receive the highest priority for treatment. Second priority should be stands that are N deficient but that also likely have one other deficiency. For example, lodgepole pine stands with pre-fertilization foliar sulphate – S levels <60 ppm are likely sulphur (S) deficient and may not respond well to N fertilization. Fertilizers applied to S deficient stands should contain both growth limiting nutrients (e.g., 35-0-0-10S). Nitrogen fertilization of lodgepole pine stands with low pre-fertilization foliar B levels (i.e., <10 ppm) may result in severe top die-back deficiency symptoms. It is recommended that stands with pre-fertilization foliar boron (B) levels <10 ppm not be fertilized or that a small amount of B (i.e., 1.5–3 kg/ha) be added to the fertilizer mix. Stands with complex nutritional problems, deficient in many nutrients, should not be fertilized.
-Screening trials
Screening trials utilize foliar analysis and small scale fertilizer trials to rapidly estimate fertilization response potential. As with foliar analysis, they should only be done when a candidate stand meets all other biological, operational, and wood supply criteria and the potential response from the treatment is not known. If a year and a half lead time is available after stand ranking is complete, consider installing some screening trials. A screening trial is used to diagnose specific nutrient deficiencies and to predict which stands will likely respond to fertilization. Screening trials may be appropriate for species with little research data or inconsistent response. Screening trials are not normally necessary for coastal Douglas-fir as sufficient predictive capabilities are now available. Western red cedar or western hemlock are not appropriate for operational screening trials because of their indeterminate buds. Research trials are necessary to obtain response data from indeterminate species. A screening trial can be initiated in the spring with response determined from foliar samples taken in the fall of the same year. The results can be used in planning the next years fall fertilization program. Screening trial methodology is described in Appendix 3.
Insects, disease, and small wildlife
The susceptibility of a stand to certain damaging agents may, or may not, be increased by fertilization. The degree of damage that can be accepted will vary by forest health factor and severity of impact. A forest health specialist should be consulted in situations where insect, disease, or animal factors may affect the priority rating of candidate stands.
Laminated root rot (Phellinus weirii) is the most significant fungal pathogen affecting growth and survival of Douglas-fir. On coastal sites, fertilization appears to have little effect on the incidence or spread of the disease. Minimally infected stands (<6% incidence of trees infected) close to rotation age may be treated. Fertilization of these stands may help them achieve harvestable size before they become severely infected. Evaluate immature Douglas-fir stands infected with laminated root rot and concentrate fertilizer operations on non-infected strata. Little is known about the effects of fertilization on the spread of other root diseases. However, N-fertilized Douglas-fir in the Intermountain region (Northern Idaho, Montana, and eastern Washington) is thought to be more susceptible to armillaria root rot (Armillaria ostoyae). Plans to treat stands that are infected with any type of root disease should be reviewed by forest health specialists.
The spruce weevil (Pissodes strobi) is the principal insect that may affect priorities for fertilizing in coastal immature Sitka spruce stands. Refer to forest health specialists for advice since increased levels of weevil attack following fertilization have been observed.
The western spruce budworm is found in parts of the Vancouver, Kamloops, and Cariboo Forest Regions. The implications of budworm infestations in relation to fertilizer programs cannot be generalized. Problems regarding specific candidate stands should be referred to forest health specialists.
In the interior, sharp increases of red squirrel feeding damage on lodgepole pine have been observed after fertilization. If any pre-fertilization damage is noted in the general area of the stand, consult regional forest health specialists and stand tending foresters for advice on whether to fertilize.
Guideline
Consult with local forest health specialists and stand tending foresters when dealing with laminated root rot, tomentosus, armillaria, white pine weevil, western spruce budworm, and red squirrel in areas being considered for fertilization treatments. Treatment may increase stand risk to damaging agents.
Operational factors
The following four operational factors should be considered during the evaluation of candidate stands.
Location: Distance to haul the fertilizer affects transportation costs. Also, costs of future commercial thinning and final harvest are partly determined by hauling distances to manufacturing plants and markets.
- Access: Conditions of access also affect costs of transporting material and personnel in fertilizer operations, in addition to later expenses of hauling timber to manufacturing plants.
- Slope: Costs of future management (e.g., commercial thinning) and harvesting usually increase as terrain becomes steeper. Furthermore, flying over steep or irregular, contoured land may not be conducive to efficient and uniform aerial distribution of fertilizer.
- Project and Block Size: Project and block sizes affect efficiency and cost of operation. Large-scale programs (e.g., >300 ha) every 2–5 years are generally more cost effective than small-scale programs every year.
Fertilizing method and application
Type of fertilizer
There are two general classes of fertilizer: organic (e.g., sewage biosolids, fish morts, secondary pulpmill effluents) and inorganic. Most operational fertilization has focused on the application of inorganic fertilizers because of their known chemical and physical properties and their cost-effective means of application.
-Organic fertilizers
Biosolids are treated waste water sludge that meet quality criteria for beneficial land application. They can act as an excellent slow-release fertilizer. Although biosolids are not currently used operationally for forest fertilization they will likely play an increasingly important role in the rehabilitation of roads, landings, and other degraded sites, as well as enhancing forest growth. A regulation is in place for the use of municipal solid waste compost. A regulation for the use of biosolids will soon be in place. Compliance with regulations is required when applying those organic fertilizers. A waste management permit must be acquired from a Ministry of Environment, Lands and Parks office prior to application of any organic fertilizer not covered by regulation.
-Inorganic fertilizers
Only four elements are currently operationally applied through use of inorganic fertilizers on forests in British Columbia. These are N (nitrogen), S (sulphur), P (phosphorus), and B (boron). Nitrogen is applied in the largest amount with S, P, and B being added to ensure the benefits of the nitrogen fertilizer are not limited by deficiencies of other elements.
Supplements of nitrogen frequently improve growth of coastal Douglas-fir, lodgepole pine, and sometimes western hemlock. Urea ((NH2)2 CO) is currently the only fertilizer widely used by the B.C. Forest Service for operational applications. This is due to its good response history, ease of storage, availability, environmental effects, and high nitrogen content (46%) which minimizes application costs per unit area.
“Forestry grade” fertilizer (approximately 3–5 mm prill diameter) should be used rather than “agricultural grade” (approximately 2–3 mm prill diameter). Aerial distribution of forestry grade prills is more uniform and the larger particles penetrate the canopy more easily.
On the coast, urea (46-0-0) is generally the only fertilizer operationally applied to Douglas-fir forests. Fertilization with N + P has been effective in young western hemlock stands growing on substrates rich in decaying wood in the CWHvh subzone and salal prone sites at low elevations in the CWHvm subzone.
In the interior, S deficiencies may limit the responsiveness of lodgepole pine to N fertilization over fairly extensive portions of the Sub-boreal Spruce and Sub-boreal Pine–Spruce biogeoclimatic zones within the central and northern portions of the interior plateau. It is recommended that S be combined with N in the operational fertilizer mix when pre-fertilization foliar sulphate-S levels are <60 ppm. A forest grade blended fertilizer (35-0-0-10S) consisting of 58% urea (46-0-0) and 42% ammonium sulphate (21-0-0-24S) is currently used in the B.C. Interior.
Guideline
The type of fertilizer (organic/inorganic) and the amount (kg/ha) of each elemental component should be identified in the silviculture prescription or stand management prescription for the site.
Rates and timing for nitrogen application
The following information only applies to forest grade urea fertilizer unless otherwise stated.
Application rates
Application rate refers to the amount of the element applied, not to the total weight of the fertilizer. Although growth increments may improve as dosage of fertilizer increases, response per unit of nitrogen is generally most cost effective for coastal areas when treatments use 200–225 kg N/ha. This rate is therefore recommended. The equivalent dose of urea prills is 435–490 kg/ha. In the interior, it is recommended to use a urea-ammonium sulphate fertilizer blended to deliver 175–200 kg N/ha and 50–60 kg S/ha.
Guideline
Any significant changes to the application rate during treatment must be verbally conveyed to the district manager followed by a written notice.
-Frequency of application
A single application of fertilizer will generally increase the growth of a treated stand for more than six years. To ensure maximum benefit from each application, and to attain the highest N efficiency (m3/kg N), treatments should not normally be repeated until at least six years have elapsed. However, more frequent applications (e.g., every 3–4 years) will be necessary if the objective is to maintain growth rates at, or near, optimal levels.
-Season of application
The season is not as important as the weather conditions under which urea fertilizer is applied. The ideal season of application is when roots are actively growing, temperatures are low, soils are wet, and precipitation is frequent. Under such conditions losses due to volatilization are greatly reduced. In community watersheds with lakes or reservoirs, do not apply fertilizer during times of rising water temperatures. Generally, do not apply fertilizer during the following periods:
- April 1–September 15 (Coast)
- May 15–September 15 (Interior: Cariboo, Prince George, and Prince Rupert Regions)
- April 15–September 15 (Interior: Kamloops and Nelson Regions)
Guideline
- Spread urea in cool and moist conditions (October–April).
- Fertilization on snow is not currently recommended if the ground is solidly frozen, the surface of the snow is crusted, or if slopes exceed 30%.
Effects on growth are generally similar for urea applications in spring compared with treatments during fall, but late fall operations (October and November) are preferred since weather is fairly predictable. For the interior, fall applications will be earlier in the north and later in the south. In the spring, temperature, time of snow melt, and consequent accessibility can vary considerably, and other tasks (e.g., planting, planning for summer work) have significant priority. Therefore, whenever possible, plan the fertilization project for the fall.
Heliports
Cost is the primary reason for contractor selection. This criteria has led to helicopter application being the main delivery system in B.C. Fixed wing aircraft require an air-strip and have limited use in British Columbia’s rugged topography. Heliport details should be worked out with the contractor. The heliports must allow for safe and efficient loading of helicopters. Remove danger trees near the heliport which jeopardize the safe operation of the helicopter. In addition, personnel involved in the operations should be briefed on working around helicopters and spectators kept at a safe distance.
When a heliport is located on a main logging road, arrangements must be made to accommodate local traffic in a manner which ensures safety for the general public as well as the application crew. Signs must be posted far enough back to give truckers plenty of warning. When large highway trucks are used to haul the fertilizer they require a turn around or loop past the heliport for the return trip. These trucks will require radio communication on active logging roads and, in some cases, pilot vehicles to escort them to the heliport.
Heliport distance from treatment area is one of the most important factors in determining cost of application. Heliports should optimally be within 1 or 2 kilometres of treatment area and if possible, at equal or higher elevation than the stands. Because of the extra concentrations of fertilizer present at a loading site and the potential for spills, all heliports should be located in dry areas, well removed from ditches or natural water bodies. Any drainage through heliports must be directed away from ditches or streams.
Guideline
- Heliports must be designed for safe and efficient operation.
- When a fertilization treatment occurs within a community watershed the heliports should be located outside of the watershed if possible.
- Heliports should be inspected for proper drainage prior to treatment and on a regular basis during treatment to clean up spills.
Pre-fertilization block layout
- Block layout is important to ensure an efficient operation while protecting other resources. Clearly marked blocks with suitable heliports close to treatment areas will minimize application time and reduce the risk of affecting other resources.
- Block boundaries, water bodies, buffer zones, and hazards should be clearly marked on aerial photographs and/or project maps for use by the pilot.
On some blocks minor boundary amendments may result in leftover fertilizer. Each grouping of blocks should include an extra area to use up excess fertilizer thus reducing the cost of transporting to another project. An approved prescription and suitable map is required for the additional area.
Guideline
Block boundaries, water bodies buffer zones, and hazards, together with the appropriate flight paths, should be clearly marked on aerial photographs and/or maps of appropriate scale for use by the pilot. The pilot applying the fertilizer should be present on a pre-treatment reconnaissance flight to become familiar with the treatment units. Before fertilizer application, the boundaries of all buffer zones should be made distinguishable from the air and flagged if necessary.
No fertilizer application zone
A 10-metre “no fertilizer application zone” or buffer zone should be left around the following water bodies:
- a fisheries lake
- any designated fishery stream (S1–S4)*
- a stream that can be identified, on a pre-flight inspection, as one observable as open water that flows into any designated fishery stream (S5, S6).*
* Riparian classes are defined in Part 10 of the Operational Planning Regulation.
A typical swath width for helicopter aerial fertilization is approximately 60 m. Fertilizer is spread 30 m on each side of the helicopter. In this case a flight parallel to the water body should have the helicopter 40 m away to maintain a 10-metre buffer strip (see Figures 1 and 2).
In community watersheds the following buffers are required:
- 10 m around a flowing stream that is observable from the air
- 100 m upstream and upslope of a community watershed intake.
Any research trials in the area should be protected from treatment. Check up-to-date district maps and the Sx trial registry for any research trials. It is also wise to check with district staff for local knowledge of any existing research trials. Ensure the organization responsible for the trial is contacted and a buffer suitable to the researcher is left.
With the encroachment of residential and farmland areas on the forest land base, suitable buffers must be established to protect the rights of the private land owner. Check the Forest Cover Atlas maps for private land in the area. Leave at least a 60-metre buffer strip around residences and a 30-metre buffer strip adjacent to lands used for agriculture. This can be modified if the residence or farmland is owned by the forest landowner or the aerial application is acceptable to the resident or landowner.
Guideline
Private land, water bodies visible from the air, and other sensitive areas should be protected as follows:
- application directly over these areas should not occur
- a buffer strip (no fertilizer application zone) is required
- a flight path parallel to all buffer strips surrounding research plots, private lands, water bodies, and other sensitive areas is required.
Contact the organization responsible for any silviculture trial or research installations to determine the size of buffer strip required.
Air operations
Aircraft and hopper must be properly equipped to ensure safety and an acceptable level of application.
- Figure 1. Strategy for laying out a buffer zone to protect a stream flowing through an area proposed for fertilization.
- Figure 2. Strategy for laying out a 10-metre buffer zone for no fertilizer application along a creek.
Guideline
- Aircraft should have electronic guidance instruments capable of ensuring even and accurate application of fertilizer over the treatment units and the avoidance of non-target areas.
- Hoppers must be equipped with a calibration system or loading equipment must be equipped with an accurate metering device to provide an accurate measure of the fertilizer.
- Hoppers must have a leak proof system with a positive shut-off device easily controlled by the pilot.
- Suitable equipment must be present to enable constant ground to air communications.
In addition to proper equipment, pilots must have demonstrated ability and experience in applying fertilizer to forest land.
Fertilizer application operations should be restricted to times of adequate daylight and visibility to ensure a safe, efficient application. The applicator must stop operations when there is inadequate daylight or weather conditions arise which could adversely affect treatment effectiveness, maintenance of buffer zones (no fertilization application), or safety of application.
Guideline
- Pilots must have demonstrated ability and experience in applying fertilizer to forest land.
- Weather and visibility conditions should not adversely affect treatment effectiveness.
- Wind can affect the uniformity of the fertilizer application. Avoid application when gusty wind conditions adversely affect treatment effectiveness or maintenance of buffer zones.
Environmental Protection
Storage and handling of fertilizer
A material safety data sheet for each type of fertilizer should be reviewed by all personnel involved in the fertilization project. These sheets must be kept on site. The data sheet lists preventive and corrective measures for storage and worker safety.
-Storage
Forest grade urea is not a controlled product under WHMIS (Workplace Hazardous Material Information System) or regulated for rail or road transportation. Because of its solubility in water, however, urea must be stored in dry containers and protected from moisture. Urea is hydrophillic and moisture will make the product go lumpy.
Security of a storage area is important to prevent cattle and wildlife from consuming urea, preventing unauthorized removal of fertilizer, and protecting equipment from vandalism. Since fertilization projects last only 4–5 days at any given site, a night watchman is usually preferable to other methods of security, such as fencing.
Guideline
- Storage areas must be secure to prevent cattle or wildlife from eating fertilizer and to avoid vandalism.
Worker safety
Where prolonged and/or repeated skin and eye contact with urea is likely to occur, workers should wear safety glasses with side shields, shirts with long sleeves, and chemical-resistant gloves. Respirators may be required to prevent overexposure by inhalation where concentrations in air exceed the occupational exposure limits. However this is unlikely unless the fertilizer is being handled in an enclosed space.
Contingency plan
Prior to commencement of projects, a contingency plan must be approved by the district manager and in place, in case of accidental spills of fertilizer.
Guideline
For areas outside of community watersheds the plan must include:
- names and telephone numbers of emergency contacts in the Ministry of Forests, and Ministry of Environment, Lands and Parks
- a water quality monitoring plan if the accidental spill occurred in a water body
- an accident clean up procedure, including remediation of the site and disposal of spill material.
Guideline
For areas inside community watersheds the contingency plan must include measures to ensure that water quality is maintained. The contents of the plan must include, but are not limited to, the following guidelines:
- names and telephone numbers of emergency contacts in the Ministry of Health; Ministry of Forests; Ministry of Environment, Lands and Parks; and the water purveyor
- plan for an alternate water supply until levels return to normal. The need for alternate supplies will be determined by the medical health officer
- water quality monitoring program to document water quality returning to background, or to a level acceptable by the Ministry of Health
- accident clean up procedures, including remediation of the site and disposal of spill material.
The spill contingency plan should be reproduced on waterproof paper and carried in designated vehicles. Ensure the contractor implementing the fertilization application is adequately equipped and trained to take remedial action in case of an accidental spill. If the fertilizer is spilled or applied in non-target areas (e.g., water sources), the contractor shall immediately notify the forest manager. Written notice must promptly follow.
An annual report on all reportable spills and the actions taken will be submitted to the district manager.
Guideline
Fertilizer applicators must have on site at all times:
- copy of a contingency plan of action for fertilizer spills
- material safety data sheets of the fertilizers being applied
- copy of the fertilization contract with appropriate site maps showing buffer areas.
Applicators should also have a copy of the silviculture prescription or stand mangement prescription.
Protection of water resource
Nutrients from forest fertilizer applications can enter water bodies through leaching, runoff, or directly when fertilizers are applied aerially. Sensitive areas are protected using buffers and limiting the area receiving the fertilizer treatment. Water quality monitoring (Appendix 4) provides a means of characterizing the background levels of nutrients in sensitive water bodies, and provides a means of confirming the effectiveness of protection measures contained in this guidebook.
Community watersheds
The definition of a community watershed is in the Forest Practices Code of British Columbia Amendment Act, 1995. Community watershed means:
the drainage area above the most downstream point of diversion on a stream for a water use that is for human consumption and that is licensed under the Water Act for
– a waterworks purpose, or
– a domestic purpose if the licence is held by or is subject to the control of a water users’ community incorporated under the Water Act
if the drainage is not more than 500 km2 and the water licence was issued before June 15, 1995.
Additional areas that do not meet this definition may be designated by a regional manager.
Community watersheds include municipal and other water user communities. Water user communities, as defined in the Water Act, have six or more licensed water users (registered with the Water Management Branch) extracting water from the same water source.
Up to 30% of a community watershed can normally be fertilized in any 12-month period. If buffer zones cannot be maintained around at least 75% of the total length of flowing streams then a maximum of 12% of the watershed can be fertilized in a 12-month period.
Do not fertilize in community watersheds if waterbodies contain high chlorophyll levels. Check waterbodies for presence of clumps or strings of periphyton (algae) attached to large stones or aquatic plants. Periphyton strings greater than 3–5 centimetres are indicative of high chlorophyll levels. Such waterbodies may exceed the chlorophyll limits for community watersheds stated in the Silviculture Practices Regulation. Sampling for chlorophyll should not be necessary if care is taken to visually assess the presence of periphyton. Sampling procedures for periphyton are contained in the Biological Sampling Manual, B.C. Ministry of Environment, Lands and Parks, Water Quality Branch. High levels of periphyton can be caused by excess nutrients in the water or the absence of benthic organisms that feed on the algae.
In addition to guidelines established in previous sections, special requirements are needed for fertilizer application in community watersheds.
Fisheries resources
Sensitive aquatic environments that are important for supporting various life history stages of fish must be protected from the direct toxic effects of fertilizer elements (e.g., ammonia, nitrite-N), and indirect effects due to eutrophication. In extreme situations, the eutrophication resulting from excess runoff of fertilizer can lead to a reduction in critical levels of dissolved oxygen necessary to sustain aquatic life, and can cause other habitat impacts. Fisheries-sensitive areas can include fish-bearing streams, tributaries that flow into fish-bearing streams, ephemeral watercourses and flood channels, swamps, seasonally flooded depressions, lake spawning areas, or estuaries.
Protection for these sensitive areas has been provided within this guidebook (see the section on Pre-fertilization block layout) principally through the use of 10-m buffer zones. Fisheries-sensitive areas containing significant numbers of streams that cannot be seen from the air are not appropriate for aerial fertilization. Streams not visible from the air cannot practically be buffered.
Cattle and wildlife
Three aspects should be considered with regard to wildlife when applying fertilizer: the availability, palatability, and inherent toxicity of the applied fertilizer. Urea applied to forest soil as forestry-grade pellets is available to wildlife for a period of a few hours to a few days, depending mainly on precipitation patterns after fertilization. Although small mammals and birds quickly lose interest in the urea pellets, larger wildlife and domestic cattle are attracted to it. Small doses (a few handfuls) of urea are lethal to these animals.
Ruminants such as deer may have a similar reaction as cattle. The prime concern is spills around the loading site.
In range use areas, arrange with the range resource officer to have cattle off the site during treatment. It is improbable that cattle will ingest a lethal dose from a properly spread fertilizer application within the block or from compacted surfaces such as roads or landings. However, cattle are attracted to urea and will even make holes in unopened bags. Therefore, do not leave fertilizer stores unattended on a site if cattle are expected to be in the area.
Daily clean up is required of loading sites to prevent fertilizer being ingested by animals.
Guideline
- Any fertilizer spills must be cleaned up on a daily basis.
Monitoring and Reporting
A fertilizing project should be monitored for compliance with any treatment standards set out in the silviculture prescription or stand mangement prescription. The following methodology is suggested for monitoring of aerial fertilizer applications. Application rate must be within 15% of target application rate.
Monitoring aerial application
To be effective, monitoring plans and procedures must be identified prior to initiating the contract. This section reviews the major points to be considered. When monitoring contractor performance:
- Check that loading sites are cleaned daily.
- Observe flight paths, flying procedures, and fertilizer spread from the bucket.
- Ensure that weather and visibility conditions do not adversely affect treatment effectiveness.
- Walk the block perpendicular to flight path to check for even coverage. Walking the block is essential to check for areas which have been missed or received too much fertilizer. Measure swath overlap to ensure the required overlap is occurring. The section on monitoring procedures gives procedures for swath overlap measurement.
- Monitor to ensure that fertilizer is not being applied outside block boundaries and in buffer strips. The boundaries and buffers must be walked to do this. The boundaries where the pilot must either turn on or off the bucket should be given particular attention.
- Keep track of the cumulative weight of fertilizer spread on the block of known area. This should normally be done by the contractor. By calculating the cumulative load weight for a given block and dividing by the known area of the block, you can determine the application rate. This calculation lets you know if the block received the correct amount of fertilizer. However, it does not confirm that this fertilizer was spread evenly. Walking the block and measuring the swath overlap are necessary to check for even spread.
Monitoring a contractor’s performance is easier if the application contract clearly identifies the standards required. A fertilization contract should detail start and finish dates, obligations of both parties, scheduling, standards of performance, payment, suspension and cancellation, fire prevention, liabilities, and operational specifications including provisions for environmental protection.
Monitoring procedures
Monitoring can normally be done by one person. If problems arise an extra person can be brought in so one person is in the landing and another in the treatment unit. Good communication with the contractor is a necessity. Monitoring must begin right away. Any necessary modifications to the application can then be worked out early with the contractor.
The uniformity of the fertilizer application is dependent on the spreader output rate, ground speed, swath width and swath overlap. The contractor should calibrate the spreader output rate and ground speed to obtain the desired coverage based on the swath width and target overlap. Monitoring procedures to check these factors are listed below.
Spreader output rate
The spreader output rate can be determined by timing how long the hopper takes to empty with a known weight of fertilizer. This should be done periodically throughout the application. The drop rate can vary with the fertilizer grade, type of equipment, and equipment wear. The spreader mechanism can be calibrated to obtain the desired spreader output rate.
Swath width
This width will vary with the spreader mechanism and grade of fertilizer. Within the range of altitudes flown during fertilization, swath width can be considered independent of altitude.
Swath width must be measured at the start of treatment to ensure proper application along buffers. Swath width can be easily measured by having the helicopter apply a swath across a logging road. With forestry grade pellets, some will be flung outside the swath. The edge of the swath is where the uniform distribution of pellets rapidly tails off to a few scattered pellets.
Ground speed
A constant ground speed is obtained by adjusting air speed to compensate for wind. For a given spreader output rate and swath width, the weight of fertilizer per area decreases with increasing ground speed. An accurate reading of ground speed is possible with aircraft equipped with a global positioning satellite system (GPS). However, using the following techniques it is not necessary for the personnel monitoring the fertilizer operation to know ground speed.
Calibration
The contractor can modify the spreader output rate and/or the speed of the aircraft to ensure that the target application rate is being obtained. This calibration should be done at the beginning of the contract. To check calibration:
- Calculate the distance over which a known weight of fertilizer should be spread to obtain the target application rate for a single swath.
- Measure out this known distance and clearly mark the start and end points so the pilot can see them.
- Have the pilot fly the line and spread the known weight of fertilizer.
- Check that the actual distance obtained provides an application rate within 10% of the target rate.
Example:
The target application rate and the swath overlap should be listed in contract specifications. The swath width can be measured as previously described.
Known:
Target application rate: 450 kg/ha
Swath width: 66 m
Swath overlap: double coverage (50% overlap)
Weight of fertilizer in hopper: 500 kg
Each swath provides half the target application rate (i.e., 450 kg/ha / 2 = 225 kg/ha). The 500 kg of fertilizer in the hopper should therefore cover 2.22 ha.
500 kg divided by 225 kg/ha = 2.22 ha or 22 222 m2 (1 ha = 10 000 m2)
With a 66-metre swath, the distance necessary to provide the target application rate can be calculated.
66 m swath width ´ swath length = 22 222 m2
swath length = 22 222 m2 divided by 66 m
swath length = 336.7 m
For this example, the actual distance flown can be between 303 and 370 metres. This will provide an application rate within 10% (plus or minus) of the target application rate for a single swath width.
If the application rate varies by more than 10% from the target, then the contractor should be responsible for further calibration. If the calibration is within 10%, then proceed with the application. During the operation, the area should be walked to check for even coverage (see Monitoring aerial applications) and the swath overlap should be measured.
Swath overlap
The pilot lays down fertilizer in parallel swaths. The second swath of fertilizer will overlap the first so that part of the ground is fertilized twice.
The aircraft must first be in the right place to deliver an accurate and consistent swath overlap. Variation from the target is likely if the flying is being done without the use of electronic guidance (e.g., transponders). The use of electronic guidance, to provide for line flying guidance should be a contract requirement.
With less than 50% overlap (double coverage), strips having double coverage alternate with strips having single coverage. To get even fertilizer distribution, there must at least double coverage over the entire area.
Measurement of coverage can only be done by standing under the aircraft during active fertilization.
Stand directly under the helicopter as it passes overhead. The fertilizer pellets will hit the ground and bounce like hailstones. Wear suitable clothing and eye protection. After they have stopped bouncing and before the aircraft returns, walk across the fertilized swath to the new outside edge where the unfertilized ground begins. Mark this edge with flagging tape. With forestry grade pellets, some will be flung outside the swath. The edge of the swath is where the uniform distribution of pellets rapidly tails off to a few scattered pellets.
On the next flight path, the aircraft will lay down another swath. As before, once the pellets stop bouncing, walk across to the new outside edge and mark it with a second piece of flagging tape. This distance is shown on Figure 3.
Figure 3. Fertilization overlap after two swaths.
Measure the horizontal distance between the two pieces of tape. Repeat this process for six consecutive flight lines to get five horizontal distance measurements. For slopes greater than 10% use a clinometer and slope tables to convert slope distance to horizontal distance. For slopes less than 10% hold the tape horizontally.
This procedure should be repeated until six consecutive edges have been identified and five consecutive horizontal distances measured. Then another location in the block should be chosen and a second sequence of six consecutive edges identified and five consecutive horizontal distances measured.
Average the overlap from the measurements. A variation of 10% or greater from the target overlap should be discussed with the contractor so further flight lines can be corrected.
Final application rate
The final check is a calculation of application rate based on the total weight of fertilizer applied and the area of the treatment unit. This check relies on the accuracy of the weighing scale used by the loader. If a check against this scale is desired, the cumulative loads for a given truck load can be checked against the Bill of Lading from the fertilizer manufacturer. The total application rate should be within 10%, plus or minus of the target application rate. Greater than 15% variation from the target application rate is considered unacceptable.
Guideline
- Measure swath width.
- Spreader output rate and/or the aircraft speed should be calibrated to ensure that the target application rate for a single swath width is achieved.
- Monitor field application to ensure there is at least double coverage.
Guideline
- Application rate must be within 15% of target application rate.
Monitoring water quality
When fertilization projects encroach on streams or water bodies, or occur in community watersheds, special precautions must be taken.
Water quality sampling
Water quality sampling is required in community watersheds and should be done when fertilizing near fisheries-sensitive zones. Samples should be taken before, during, and after treatment. Guidelines for sampling and analysis are provided in Appendix 4.
Guideline
- Water quality sampling is required in community watersheds and should be done when fertilizing near fisheries-sensitive zones.
Reporting of accomplishments under SMPs
Reporting of stand management prescription accomplishments on free growing stands is required for major licence holders.
The Silviculture Practices Regulation requires that a treatment report is submitted quarterly to the district manager. This report must:
- include a summary of the silviculture treatments, including fertilization, accomplished on specified areas. Fertilization information should include product type, amount (kg/ha) of fertilizer applied, and a location map
- cover the period beginning two weeks before the last report date and ending two weeks before the current report’s submission date
- be signed and sealed by a professional forester.
The submission dates are April 15, July 31, October 31, and January 15.
Appendix 1: Stand selection guidelines for use in stand management prescriptions
These stand selection guidelines are intended to aid in the evaluation and selection of appropriate sites and fertilizing treatments for inclusion in stand management prescriptions. The guidelines are ranked from most desirable (1) to least desirable (3) with some stands ranked as treatment Not Recommended (NR).
When assigning priority to a particular stand, as many factors as possible should be considered. Stands having average rankings of high or medium will receive greater preference for fertilizing, while areas with low rankings will be considered less suitable for fertilizing.

Appendix 2: Foliar sampling guidelines
Foliar analysis is used to indicate whether fertilizer may result in a growth response on a particular site. Foliar analysis is not necessary for all stands. It should only be necessary where analysis of other location, site, and stand factors suggest that nutrients other than N may limit fertilizer response (see Table 1 in the section on biological factors). The analysis must be scheduled so that there is sufficient time to order nutrient supplements if indicated by the foliar analysis. Check with the lab well in advance to ensure samples can be analyzed within your time frame. This appendix provides guidelines for foliar sampling and a brief description of sampling methods. For more extensive coverage, refer to Land Management Report No. 20, Evaluating Forest Stand Nutrient Status (Ballard and Carter, 1985).
Foliar sampling guidelines
For a relatively homogeneous stand type, set up two or more well separated transect lines that cross the stand and any major topographic features. Exclude portions of the lines which may be contaminated by dust (guideline 4 below). Establish equally spaced reference points on these lines, corresponding to the number of trees to be sampled. From each reference point, select the nearest tree that meets guidelines 2 and 3 (below).
For non-homogeneous blocks, use stratified sampling. Where a stratum occurs as numerous small “islands” within the stand, use the above transect approach within the island, to identify individual trees for sampling. This amounts to sampling each stratum as if it were an individual stand. If a stratum occurs as two or more large islands, it is appropriate to use one transect line in each of two (or more) of the largest islands.
Use the following guidelines:
- Sample conifer foliage during the dormant season, preferably between September 15 and December 15.
- Confine sampling to dominant and co-dominant trees.
- Avoid trees likely to yield poor samples because of heavy cone production, insect, or disease damage.
- Avoid sampling foliage near unpaved roads or in other situations that may lead to foliage contamination from dust.
- Estimate the distance from the top to bottom of the live crown. Collect samples from the top 1/4 to 1/2 of the live crown excluding the top 3 whorls. Sample the current year’s foliage from two branches per tree by collecting a 2 g (fresh mass) subsample (terminals and/or laterals) from each branch.
- Usually it is best to put samples into labeled plastic bags, although samples collected under dry conditions can be put into labeled paper bags. Dirty samples should be discarded and replaced.
- Drying of samples should begin as soon as possible, preferably on the day of sampling. However, if samples are kept cool (1–5°C), drying may be deferred as much as a week and perhaps longer. It may be easiest to ship samples directly to the analytical laboratory if prompt transport and handling can be arranged. If samples must be dried before shipment, oven drying is best. If no oven drying facilities are available, thorough air drying should be done.
Use color and size of needles to differentiate between current and previous year foliage. The color of foliage is often different for different foliage ages, and can vary from stand to stand. Foliage from the current year is often paler or more yellow-green. Twigs are invariably darker on shoots from the previous year than on shoots from the current year, as are fascicle sheaths in pines. Twig diameter is usually larger in terminal shoots that developed during the previous year and the terminal buds of such shoots, if they have failed to flush, are often larger in diameter and somewhat longer than current-year buds. Care should also be taken to avoid sampling lammas growth.
Dry the samples by transferring them to short, fully opened paper bags (e.g., lunch bags, with the top half rolled down or cut off) on which the sample number has been written. Before drying, remove and discard twigs, bud scales, and other non-foliage material (fascicle sheaths are not normally removed from pine needles). When air drying, leave the bags at ordinary room temperature and humidity for several days. It is preferable to use an electric fan for constant circulation when air drying. When oven drying, use a temperature of 70°C for 8–12 hours, or until needles snap cleanly in two when bent. After drying, fold the top of the bag over and staple shut.
Microwave ovens can also be used to dry foliage samples. Samples should be in paper lunch bags that have the tops cut off sufficiently to allow the bags to stand up inside the microwave. Turn the oven on to maximum power for 2–3 minutes, open the door and check the samples. Then use 2-minute bursts of heat, opening the door to let the steam escape, and 1-minute shots as the samples dry. Check the needles between bursts of heat and mix them up in the bag a little to even out the drying process. The total drying process will take about 8–12 minutes. Needles will burn if over-dried, so be careful as they dry out. The foliage is ready for shipping if it snaps when bent.
Wet foliage can pick up sulphur and boron from paper bags, but slightly damp foliage won’t be at much risk. Samples may be collected in the field in paper bags and, after drying, moved to labelled Ziploc bags for shipment to the lab. Samples collected in heavy rain, or those covered in ice or snow, should be collected in plastic bags, allowed to air dry inside, and then put in paper bags for drying.
If foliage samples are to be used for the evaluation of treatment impacts associated with fertilizer screening trials or other silvicultural treatments it is important to determine needle weight prior to grinding. Ensure that the laboratory is made aware of this requirement where necessary.
Foliage sampling methods
Foliage samples may be collected from the ground by climbing or shooting, or by helicopter. The method chosen depends on tree height and form and the extent of the sampling program. Foliar sampling can efficiently employ two persons: one collecting while the other separates the growth of the current year, bags the samples, indicates when a sufficient sample has been collected from a tree, labels bags, and records field notes.
Ground sampling
If trees are less than 10 m tall, samples may be collected with a hand pruner, long handled orchard pruner, or telescopic pruning pole.
Climbing
Climbing is most feasible in stands less than 30 m in height. See Land Management Report No. 20 for a complete description.
Shooting
Shooting down samples is often the only practical alternative in forest stands greater than 20 m in height. If properly conducted, it can be less hazardous than climbing for samples. See Land Management Report No. 20 for a complete description of the procedure including selection of guns, ammunition and licensing requirements.
Helicopters
If foliar samples are to be collected from several stands in close proximity to each other it may be most efficient to sample from helicopter with a certified helicopter operator.
Table 1. Interpretation of macronutrient concentrations in current year’s foliage of five commercial conifer species of the Pacific Northwest. (From: Diagnosis and Interpretation of Forest Stand Nutrient Status, Carter, R. In Forest Fertilization Sustaining and Improving Nutrition and Growth of Western Forests. H.N. Chappell, G.F. Weetman, R.E. Miller (eds.). 1992.)
Table 2. Interpretation of micronutrient concentrations in current year’s foliage.1 (From: Diagnosis and Interpretation of Forest Stand Nutrient Status, Carter In Forest Fertilization Sustaining and Improving Nutrition and Growth of Western Forests. H.N. Chappell, G.F. Weetman, R.E. Miller (eds). 1992.)
Appendix 3: Guidelines for screening trials
Introduction
First-year increases in needle weight and foliar nutrient concentration can provide a useful quick index of long term stemwood growth response to fertilization. As such, fertilizer screening trials can be established in candidate stands to predict which stands are likely to respond to fertilization. If established in the spring, the first year foliage response can be obtained in the fall of the same year. The needles of fertilized and unfertilized trees are weighed to find if there has been an increase in needle weight as a result of fertilization. Foliar analysis will show any change in the nutrient status of the trees associated with the fertilizer application. The results can then be used to rank candidate stands on the basis of fertilization response potential and to finalize plans for the next year’s fall operational fertilization program.
Screening trials should only be established after a candidate stand meets all other biological, operational, and wood supply tests. Screening trials are most appropriate for species with little fertilization research data or inconsistent growth response.
Criteria
The predictive reliability of screening trials is dependent on how well the changes in needle weight and foliar nutrient concentration reflect subsequent stemwood response. As such, four important criteria must be met:
- The species examined must be determinate (i.e., leaf primordia are initiated in the previous year).
- Only the current year’s foliage is used in the analysis.
- The methods used for collecting, handling, and drying foliage samples are consistent with those outlined in Appendix 2 and in Land Management Report No. 20.
- The trees selected for sampling are representative of the stand being considered for fertilization.
If these criteria are met, the following guidelines can be used to establish screening trials within candidate stands.
Mini-plot establishment
Within the candidate stand, two adjacent dominant or codominant trees with room for crown expansion should be selected for each of 15 mini-plots. The paired trees should all have similar diameters, heights, and live crown lengths, and should be growing in areas of the stand that are relatively homogeneous in terms of understorey vegetation, microsite position, soil, and stand structure. Selected trees should have no obvious symptoms of insect or pathological activity and should not have heavy cone crops.
A 0.01 ha mini-plot should be centred around each pair of sample trees and plot boundaries identified with flagging. A minimum distance of 5 m should separate the outer boundaries of adjacent mini-plots. Both of the sample trees within each mini-plot should be clearly marked with paint and flagging. Prepare a map showing the location of the 15 mini-plots within the candidate stand.
-Fertilizer measurement and application
Fertilizer screening trials normally include three treatments:
- control (i.e., unfertilized)
- N alone
- complete nutrient blend (e.g., NPKSMgB).
This makes it possible to evaluate the foliar response to N alone as well as any additional response associated with the application of other growth-limiting nutrients. In most cases, it is possible to identify which of the additional nutrients included in the blend is responsible for the incremental response. In the event of a larger growth response with the complete blend, the predicted extra response will have to be weighed against the additional costs of adding other nutrients to the operational mix.
The following fertilizers and rates are recommended for use in screening trials:
N – urea (46-0-0) @ 200 kg N/ha
P – triple superphosphate (0-45-0) @ 100 kg P/ha
K – sulphate potash magnesia (0-0-22-11Mg-22S) or muriate of potash (0-0-60) @ 100 kg K/ha
Mg – sulphate potash magnesia or magnesium sulphate (0-0-0-10.5Mg-14S) @ 50 kg Mg/ha
S – sulphate potash magnesia or magnesium sulphate @ 100 kg S/ha
Micronutrients (e.g., Cu, Zn,) can also be added to the complete blend where deficiencies are suspected.
The specific nutrients to be included in the complete blend and the amount of each actual nutrient to be added per hectare should be discussed with the fertilizer supplier prior to ordering. The calculated amounts of fertilizer to be applied per mini-plot should be carefully checked prior to application.
The amount of fertilizer to be applied to each 0.01 ha mini-plot should be pre-weighed and bagged. Each of the two fertilizer treatments (N alone and complete blend) will be applied to five of the mini-plots. To ensure even fertilizer distribution, the fertilizer for each mini-plot should be divided into two equal portions with each portion being spread evenly over one-half of the plot. The five control mini-plots are left untreated.
Foliage sampling and analysis
Foliage samples will be collected from both trees in each mini-plot in the fall after fertilization. The collection, handling, and drying of foliage samples must be consistent with methods outlined in Appendix 2 and in Land Management Report No. 20.
The analysis of the foliage samples can be done by a number of commercial laboratories in the Pacific Northwest. The analysis must include determination of the weight of 100 needles (or fascicles for lodgepole pine) for each of the 30 sample trees (two trees per mini-plot). An equal weight of foliage from each of the two trees per mini-plot should then be combined to form a single composite sample for chemical analysis. After grinding, the 15 composite samples will be analyzed for all macronutrients (N, P, K, Ca, Mg, S) and micronutrients (Fe, Cu, Zn, Mn, B). Because it is particularly useful in diagnosing S deficiencies, foliage should also be analyzed for sulphate-S.
Interpretation of results
The interpretation of the needle weight results is quite straightforward. The average increases in first year needle weight of fertilized plots relative to the control will be used to assess the fertilization response potential of the candidate stand. Those stands with the largest increases in needle weight probably have the highest stemwood growth response potential. Differences in needle weight response between the N only and complete blend treatments will give an indication of the likelihood of other nutrient deficiencies.
The interpretation of the foliar data is considerably more complicated. The direction and magnitude of shifts in first-year foliar nutrient concentration, nutrient content, and needle weight are used to make diagnostic interpretations about the nutritional status of foliage with respect to both added and non-added nutrients. In cases where the complete blend gives a larger needle weight response than N alone, the nutrients(s) responsible for the extra response must be identified. A forest nutrition specialist should be consulted to help interpret foliar data.
Appendix 4: Water sampling guidelines for community watersheds and designated fish streams
Water quality monitoring
Water quality monitoring before and after broadcast forest fertilization is recommended in all community watersheds and recommended in contentious or fisheries-sensitive areas planned for fertilizer applications. In sensitive areas such as salmon spawning streams, changes in water chemical composition may negatively impact biological productivity. If there are any doubts whether water sampling should be conducted, please contact the district silviculture resource officer, B.C. Ministry of Forests for recommendations. The following guidelines are adapted from the Community Watershed Guidebook: Forest Fertilizers. These guidelines are recommended for water quality monitoring:
A. Sample sites
Choose 2 sampling sites, one upstream from the treatment area serving as the control and the other immediately downstream of the treatment area. Sampling in these locations should provide an indication of the maximum concentrations of chemicals due to fertilizer application. More sites should be selected if the treatment area covers a large area.
Sample sites should be located accurately. Only if the same location is consistently sampled can temporal changes in water quality be interpreted with confidence. Use accurately written station location descriptions, mark the sites in the field with flagging, and locate them on maps.
B. Sampling procedures
Use clean bottles and obtain water samples as close to mid-stream and mid-depth as practically possible. If sampling in a fish stream, take the water temperature at the sampling location. Temperature is necessary to assess ammonia levels. Label the bottles with the date, site location, and water temperature (if necessary).
Samples must be shipped on the day of collection in a cooler packed with ice, in order to maintain a sample temperature of approximately 4°C. The samples must be received by the laboratory within 72 hours of collection.
C. Sample frequency
Pre-application monitoring should be conducted for two weeks. Obtain three samples, twice per week at both control and treatment area sites. Obtain results back from the laboratory in a timely manner to enable changes to be made in the planned treatments if the analysis shows high levels of natural nitrogen or phosphorus.
Post-application monitoring should be conducted immediately following fertilizer application and then twice per week for three weeks. Also, if a storm event occurs during the three weeks post-treatment period, obtain water samples within 24 hours of the event. This will provide an indication on increased nutrient inputs through direct application to streams or through accidental spills.
Collect at least one sample approximately three months after fertilizer application, to document water quality returning to background levels. In the interior of British Columbia, at least one sample should also be collected immediately after spring floods.
D. Laboratory analysis
For both community watersheds and fish-sensitive streams, total dissolved phosphorus should be analyzed if phosphorus was present in the fertilizer mix. Samples must be shipped on the day of collection, and results received back from the laboratory as quickly as possible, preferably within 3 days.
--Community watershed
Pre- and post-monitoring samples should be analyzed for pH, total nitrogen, total ammonia, and nitrate-N.
--Fish streams
Pre- and post-monitoring samples should be analzed for pH, total ammonia and nitrite-N.
E. Analysis of concentrations
--Community watershed
Post-treatment monitoring results exceeding levels specified in this guidebook should be reported promptly to the Ministry of Health; the water purveyor; Ministry of Environment, Lands and Parks; and the district silviculture resource officer, Ministry of Forests. These results should then be used to guide future applications.
--Fish streams
Pre-treatment results that exceed the following thresholds for ammonia or nitrite-N are not recommended areas for aerial fertilization. Post-treatment results that exceed the threshold levels should be reported to the district silviculture resource officer, Ministry of Forests, and the Department of Fisheries and Oceans (DFO).
Nitrite-N should not exceed 0.06 milligrams/litre.
Recommended maximum levels for total ammonia (NH3) (from CCREM, 1987)
The application of phosphorus-based fertilizer should not result in detectable elevations of total phosphorus over background stream concentrations when appropriate streamside buffer zones are used. Detectable elevations should be reported to the district silviculture resource officer, the regional environmental section head for the MoELP, and the DFO.
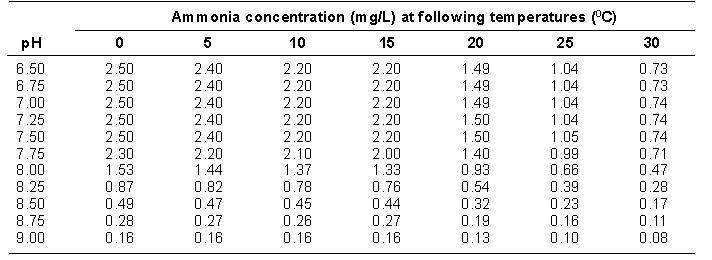
Figures
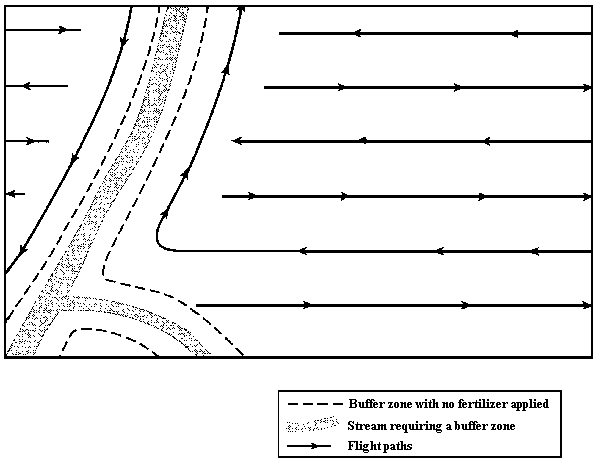
Strategy for laying out a buffer zone to protect a stream flowing through an area proposed for fertilization
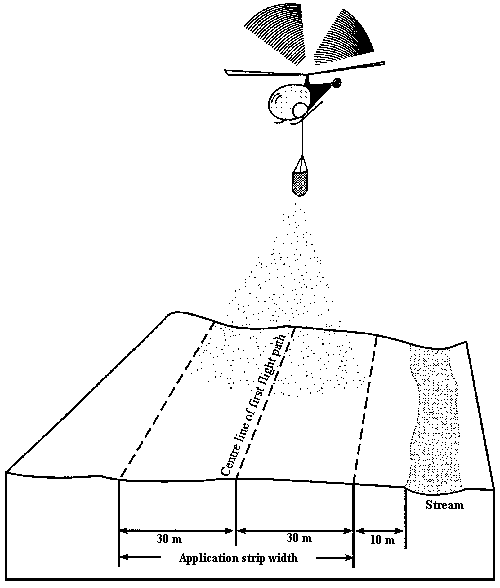
Strategy for laying out a 10-metre buffer zone for no fertilizer application along a creek
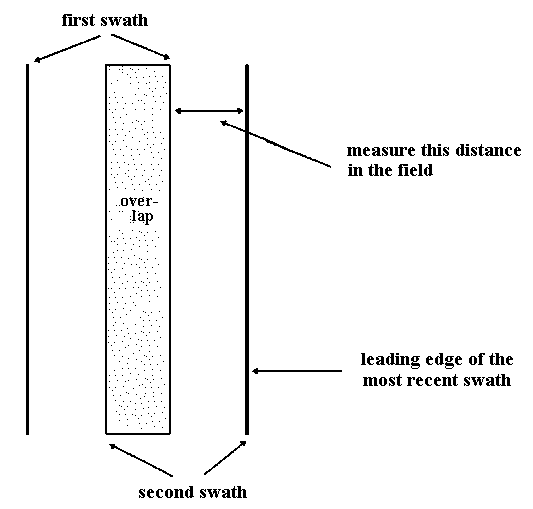
Fertilization overlap after two swaths
